The immune system is our primary defense tool against infections and various diseases, including cancer. Its power is undeniable, and when activated uncontrollably, it can cause irreversible damage to vital organs and, in many cases, lead to death. This is why various molecular mechanisms, known as immune checkpoints, exist to regulate immune activity and prevent collateral damage. However, in cancer, this can be a double-edged sword, as some tumors have “learned to hide” from the immune response by using these same mechanisms to their advantage.
The most well-known immune checkpoints today include PD-1/PD-L1, PD-L2, and CTLA-4/B7 interactions. However, in recent years, a promising candidate has emerged: HLA-G, a protein whose natural function is to maintain immune tolerance during pregnancy, preventing the maternal immune system from attacking the fetus as a “foreign entity.” This same protein can become an ally for tumors, helping them evade destruction by the immune system.
What Is HLA-G and Why Is It Important?
HLA-G (Human Leukocyte Antigen-G) is part of the Major Histocompatibility Complex (MHC) family, which regulates immune responses. Unlike other MHC molecules, HLA-G has a primarily immunosuppressive role, meaning it reduces the activity of key immune cells, including helper and cytotoxic T cells, dendritic cells, macrophages, B cells, and natural killer (NK) cells—all essential components of the antitumor immune response.
Certain immune cell receptors, such as ILT-2, interact with HLA-G, triggering a signaling cascade that silences the immune response, allowing tumors to escape detection.
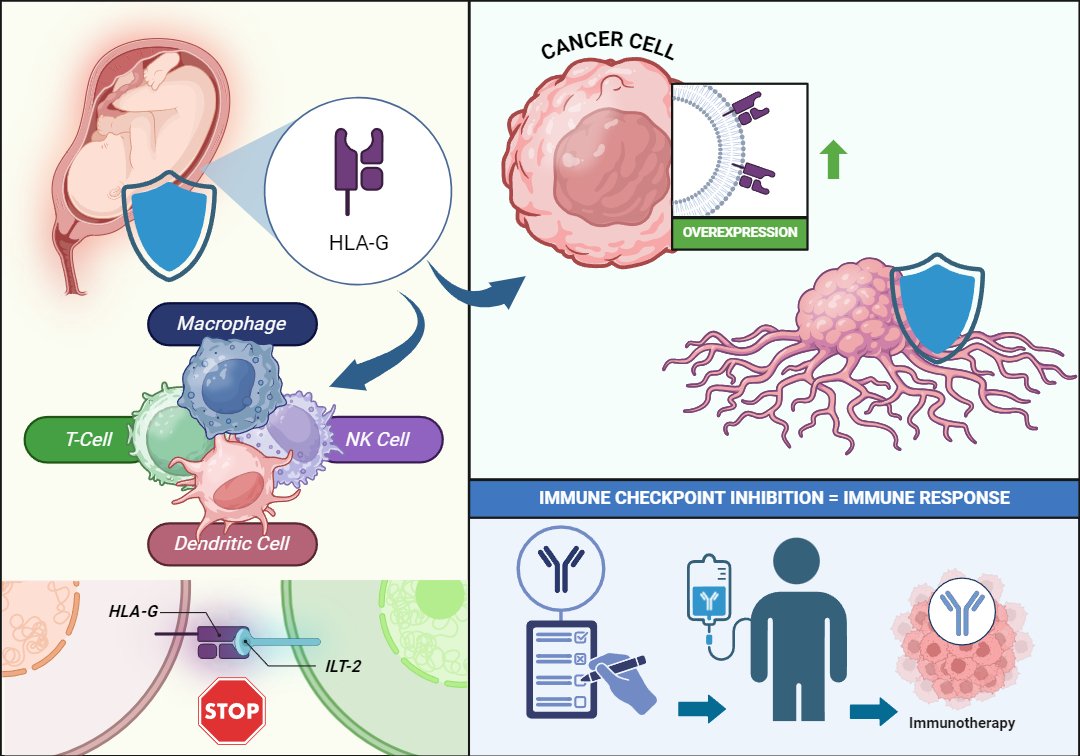
Figure 1. The Role of HLA-G in Cancer: This illustration shows how HLA-G, a protein that regulates immune responses during pregnancy, is exploited by cancer cells to evade the immune system. Immunotherapy can block this mechanism, restoring the immune system’s ability to attack the tumor.
HLA-G and Cancer: An Immune Evasion Strategy
Research has shown that some types of cancer express HLA-G, including:
- Breast cancer
- Lung cancer
- Colon cancer
- Ovarian cancer
- Melanoma
By producing HLA-G, tumors **reduce the immune system’s ability to attack them**, creating a protective shield that prevents immune cells from recognizing and eliminating cancerous cells.
Could HLA-G Be a Target for Immunotherapy?
Advancements in immunotherapy have demonstrated that blocking immune checkpoints like PD-1/PD-L1 and CTLA-4 can significantly enhance the immune system’s ability to fight cancer. This raises a crucial question: Could HLA-G be the next target for immunotherapy?
Evidence Supporting HLA-G as an Immune Checkpoint:
- Detected in multiple cancer types through immunohistochemistry (IHC) and RNA analysis.
- HLA-G-loaded exosomes (extracellular vesicles) have been identified, which suppress the immune response even in distant areas of the tumor.
- Experimental models show that blocking HLA-G with antibodies restores immune cell cytotoxicity, suggesting its role as a key mechanism in tumor immune evasion.
Challenges in Using HLA-G as a Therapeutic Target:
Heterogeneous expression—Not all studies find high levels of HLA-G in tumors, indicating variations between cancer types.
Dual role in cancer—In some cases, high HLA-G levels are linked to better prognosis, especially in ovarian and rectal cancer, suggesting its function may depend on the context.
Multiple isoforms—HLA-G exists in seven isoforms (Membrane-bound: HLA-G1, G2, G3, G4 Soluble: HLA-G5, G6, G7). It remains unclear whether all isoforms contribute equally to immune evasion.
HLA-G: A Promising Candidate, But Many Questions Remain
While studies suggest that HLA-G plays a crucial role in tumor immune evasion, several key questions must be addressed before considering its blockade as an immunotherapy strategy:
- Which HLA-G isoforms are most relevant in cancer?
- How does its expression vary among different tumors?
- What impact does it have on cancer progression and response to other immunotherapies?
HLA-G could represent a new frontier in cancer immunotherapy, but before developing treatments targeting this protein, it is crucial to understand how, when, and in which cancer types it plays a critical role in immune evasion.
HLA-G: Cancer’s Ally or Future Therapeutic Target?
HLA-G is emerging as a potential new immune checkpoint in cancer, acting as a mechanism that helps tumors evade the immune system. If its role in immune evasion is confirmed, blocking HLA-G could become a new therapeutic strategy to enhance the effectiveness of immunotherapy.
However, due to its complexity and variable expression, further studies are needed to define its true impact on cancer progression. As research advances, HLA-G could either become a key piece in the fight against cancer or prove to have a more nuanced role than previously thought.
In the era of personalized immunotherapy, understanding each component of the immune puzzle is essential for developing more effective and targeted treatments. HLA-G might just be one of those missing pieces.
Did you find this topic interesting? Share your thoughts and stay updated on the latest advancements in cancer immunotherapy!
Main Reference:
Krijgsman, D., Roelands, J., Hendrickx, W., Bedognetti, D., & Kuppen, P. J. K. (2020). HLA-G: A New Immune Checkpoint in Cancer?. International journal of molecular sciences, 21(12), 4528. https://doi.org/10.3390/ijms21124528
Other References:
Kovats, S., Main, E. K., Librach, C., Stubblebine, M., Fisher, S. J., & DeMars, R. (1990). A class I antigen, HLA-G, expressed in human trophoblasts. Science (New York, N.Y.), 248(4952), 220–223. https://doi.org/10.1126/science.2326636
Rouas-Freiss, N., Gonçalves, R. M., Menier, C., Dausset, J., & Carosella, E. D. (1997). Direct evidence to support the role of HLA-G in protecting the fetus from maternal uterine natural killer cytolysis. Proceedings of the National Academy of Sciences of the United States of America, 94(21), 11520–11525. https://doi.org/10.1073/pnas.94.21.11520
Shih, K., Arkenau, H. T., & Infante, J. R. (2014). Clinical impact of checkpoint inhibitors as novel cancer therapies. Drugs, 74(17), 1993–2013. https://doi.org/10.1007/s40265-014-0305-6


