Introduction
Immunotherapy has revolutionized the treatment of advanced melanoma, particularly with the use of PD-1 inhibitors. However, the low response rate remains a clinical challenge. A recent study published in Cell Death & Disease suggests that α-ketoglutarate (α-KG) supplementation may be key to improving the effectiveness of these treatments through epigenetic mechanisms.
In this article, the researchers In this article, Liu and a team of researchers explore how α-KG modulates PD-L1 expression and strengthens the immune response against melanoma, offering a promising new therapeutic strategy.
What is α-Ketoglutarate and Why is it Relevant in Cancer?
α-Ketoglutarate (α-KG) is a crucial metabolite in the tricarboxylic acid (TCA) cycle, also known as the Krebs cycle. Its role extends beyond energy production, as it also regulates epigenetic processes by acting as a cofactor for TET (ten-eleven translocation) enzymes, which are responsible for DNA demethylation.
This epigenetic mechanism is critical because alterations in DNA methylation have been linked to cancer progression and response to immunotherapy.
Study Methodology
To evaluate the impact of α-KG on melanoma immunotherapy, researchers conducted experiments using murine models and melanoma cell lines. Groups of mice were administered α-KG in combination with PD-1 inhibitors, while control groups received standard treatments. Throughout the study, tumor progression, immune cell infiltration, and the expression of immune response-related genes were measured.
Additionally, epigenetic studies were performed to analyze the activity of TET2/3 enzymes, responsible for DNA demethylation, and their effect on PD-L1 regulation. Techniques such as immunohistochemistry and flow cytometry were used to assess changes in the tumor microenvironment, including CD8+ T cell infiltration and M2 immunosuppressive macrophages. These experimental approaches provided a comprehensive evaluation of the impact of α-KG on the anti-tumor immune response.
How Does α-KG Improve Anti-PD1 Immunotherapy?
The study demonstrated that patients and animal models responding to anti-PD1 therapy exhibited higher α-KG levels compared to non-responders. Based on this observation, researchers explored whether α-KG supplementation could enhance immunotherapy.
One of the most significant findings was that α-KG treatment promoted PD-L1 expression in tumor cells through the activation of TET2/3 enzymes. This activation facilitated epigenetic modifications, increasing DNA accessibility in the PD-L1 gene promoter, thereby allowing greater binding of STAT1 and STAT3 transcription factors. As a result, tumor cells expressed higher levels of PD-L1 in response to IFNγ signaling, paradoxically improving the effectiveness of PD-1 blockade in murine models.
Additionally, mice treated with α-KG and anti-PD1 showed:
– Increased infiltration of CD8+ T cells.
– Reduced presence of M2 immunosuppressive macrophages.
– Significant improvement in survival rates compared to control groups.
These results suggest that α-KG not only enhances tumor immunogenicity but also modulates the tumor microenvironment to support a more effective immune response.
Molecular Mechanism:
The IFNγ-STAT1/3-CD274 Pathway One of the study’s most relevant findings was the identification of the IFNγ-STAT1/3-CD274 pathway as a mediator of α-KG effects. It was found that α-KG promotes the activation of STAT1 and STAT3, which in turn regulate PD-L1 expression in tumor cells.
This mechanism suggests that α-KG enhances immunotherapy by facilitating PD-L1 expression in a context where T cells can still recognize and attack the tumor.
Why is PD-L1 Modulation Important?
PD-L1 is a key protein in immune system regulation. Its overexpression in tumors can lead to immune evasion, but in the context of anti-PD1 therapy, its proper regulation can enhance treatment efficacy.
The study suggests that α-KG not only increases PD-L1 expression but does so in a way that enhances immune activation, avoiding a paradoxical resistance effect.
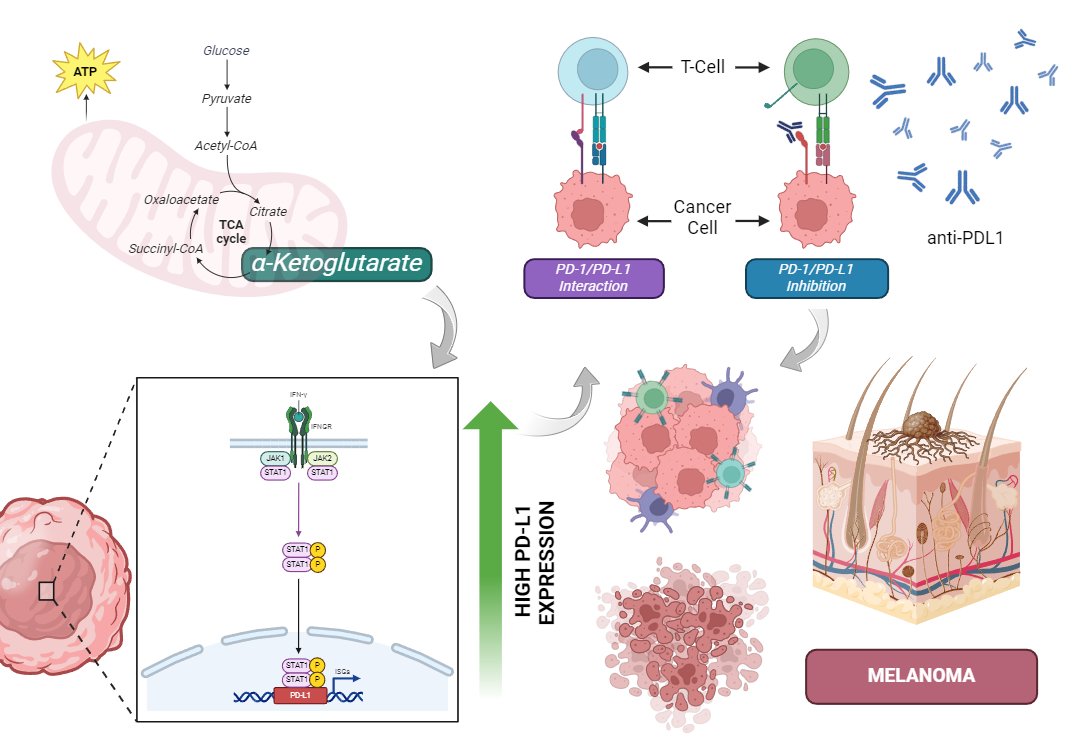
Figure 1. α-Ketoglutarate (α-KG) promotes PD-L1 expression in tumor cells via the IFNγ-JAK/STAT pathway, enhancing the response to PD-1/PD-L1 checkpoint inhibitors in melanoma.
Clinical Implications and Future Research
The findings of this study open new possibilities for optimizing melanoma immunotherapy:
α-KG could be used as an adjuvant therapy in patients with advanced melanoma, facilitating a better response to immunotherapy. Moreover, identifying epigenetic biomarkers may help predict which patients would benefit the most from this strategy. As research progresses, it will be important to assess α-KG’s effect in other cancer types that respond to immunotherapy, potentially expanding its clinical applications.
Although preclinical results are promising, clinical trials in humans are essential to confirm the safety and efficacy of α-KG in combination with PD-1 inhibitors. The clinical validation of these findings could pave the way for a new therapeutic approach based on cancer metabolic and epigenetic modulation.
Conclusion
This study highlights the key role of metabolism and epigenetics in cancer immunotherapy. α-KG supplementation could be an innovative strategy to improve PD-1 blockade efficacy in melanoma, opening new perspectives in precision oncology
As immunotherapy research advances, understanding how metabolic modulation can optimize current treatments and improve patient responses will be essential.
Main Reference:
Liu, N., Zhang, J., Yan, M., Chen, L., Wu, J., Tao, Q., Yan, B., Chen, X., & Peng, C. (2023). Supplementation with α-ketoglutarate improved the efficacy of anti-PD1 melanoma treatment through epigenetic modulation of PD-L1. Cell Death & Disease, 14(2), 1–13. https://doi.org/10.1038/s41419-023-05692-5
Other References:
Pavlova, N. N., & Thompson, C. B. (2016). The Emerging Hallmarks of Cancer Metabolism. Cell metabolism, 23(1), 27–47. https://doi.org/10.1016/j.cmet.2015.12.006
Pan, M., Reid, M. A., Lowman, X. H., Kulkarni, R. P., Tran, T. Q., Liu, X., Yang, Y., Hernandez-Davies, J. E., Rosales, K. K., Li, H., Hugo, W., Song, C., Xu, X., Schones, D. E., Ann, D. K., Gradinaru, V., Lo, R. S., Locasale, J. W., & Kong, M. (2016). Regional glutamine deficiency in tumours promotes dedifferentiation through inhibition of histone demethylation. Nature cell biology, 18(10), 1090–1101. https://doi.org/10.1038/ncb3410
TeSlaa, T., Chaikovsky, A. C., Lipchina, I., Escobar, S. L., Hochedlinger, K., Huang, J., Graeber, T. G., Braas, D., & Teitell, M. A. (2016). α-Ketoglutarate Accelerates the Initial Differentiation of Primed Human Pluripotent Stem Cells. Cell metabolism, 24(3), 485–493. https://doi.org/10.1016/j.cmet.2016.07.002