Immunotherapy has demonstrated remarkable efficacy in treating various types of cancer, achieving long-lasting responses and, in some cases, complete remissions. Its success lies in its ability to inhibit immune suppression mechanisms that allow tumor cells to evade immune attack. Among the most widely used strategies are immune checkpoint inhibitors, such as monoclonal antibodies targeting PD-1 (programmed death-1) and its ligand PD-L1, which reactivate T lymphocytes against cancer cells.
Despite these advances, not all patients respond uniformly. In some cases, a mixed response is observed, where certain tumors shrink while others remain unchanged or even progress. This response pattern suggests the existence of additional factors that influence treatment efficacy within each tumor in the same patient.
In a study published by Morinaga et al. (2022) this issue was addressed using whole-exome sequencing (WES), single-cell analysis (scRNA-seq), and preclinical mouse models. The findings indicate that intratumoral heterogeneity and variability in T-cell infiltration play a crucial role in determining immunotherapy effectiveness and patient prognosis.
Tumor Heterogeneity and Differential Immune Response.
To investigate the causes of the mixed response, two metastatic lymph node lesions from a patient with advanced melanoma were analyzed. Despite being located in the same anatomical region, they exhibited significant differences in genetic composition and tumor microenvironment.
The first lesion displayed high tumor mutational burden (TMB) and a strong infiltration of exhausted CD8+ T cells. These lymphocytes, although dysfunctional, can be reactivated by PD-1 inhibitors, explaining the tumor’s regression following immunotherapy. Additionally, this lesion contained a higher number of neoantigens, altered proteins generated by tumor mutations, which can trigger a specific immune response against cancer cells.
In contrast, the second lesion had a lower mutational burden and fewer activated T cells, indicating a cold tumor microenvironment (cold TME). In these tumors, the lack of immune cell infiltration reduces the effectiveness of immunotherapy, as there are not enough T cells within the tumor to be reactivated.
These findings reinforce the idea that tumor heterogeneity is not limited to genetic differences but also affects the tumor’s interaction with the immune system, explaining why some lesions respond to immunotherapy while others remain resistant.
Validation in Preclinical Models
To confirm these findings in an experimental setting, tumor clones derived from the same cell line were developed in mice. Two distinct populations with differential responses to immunotherapy were identified:
- Tumor clone with high T-cell infiltration and a positive response to anti-PD-1 therapy. This clone had a hot TME, characterized by abundant CD8+ T-cell infiltration, which facilitated treatment response.
- Tumor clone with a cold TME and resistance to anti-PD-1 therapy. This clone exhibited low MHC-I expression and fewer infiltrating T cells, reducing the effectiveness of immunotherapy.
The preclinical model confirmed that immunotherapy response depends not only on tumor mutations but also on the composition of the tumor microenvironment and the immune system’s ability to infiltrate the cancerous tissue.
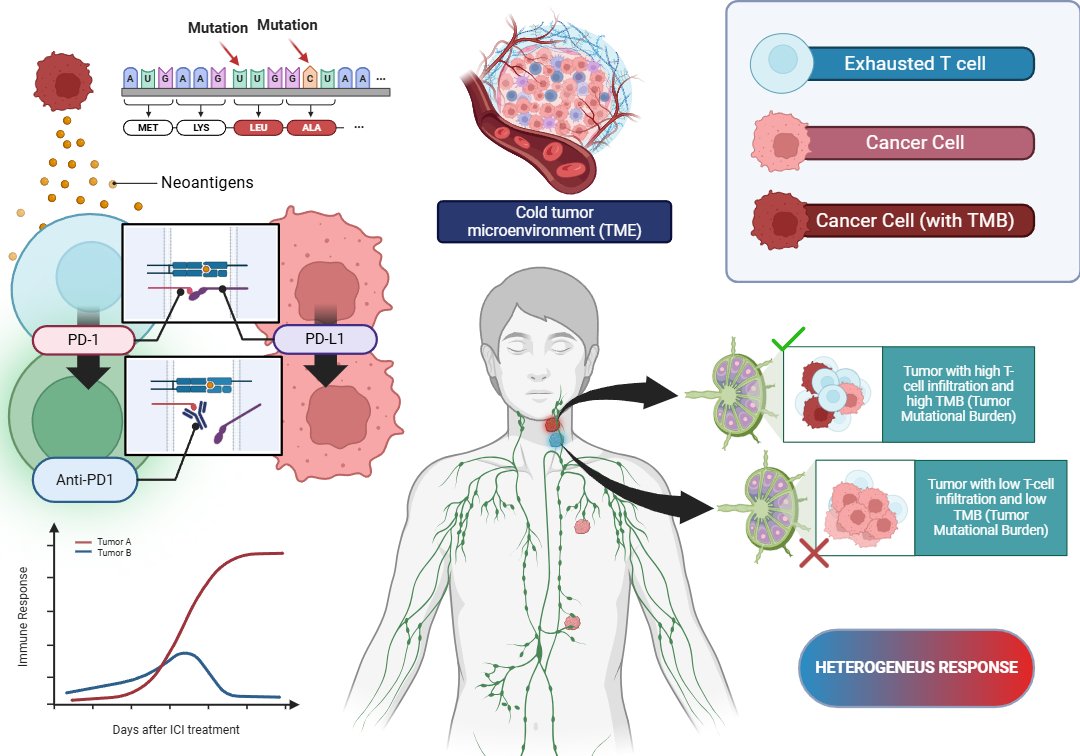
Figure 1. Tumors with high T-cell infiltration and TMB respond, while those with low infiltration remain resistant, leading to a mixed response.
Clinical Impact and Therapeutic Options
The analysis of 503 cancer patients treated with PD-1 inhibitors revealed that those with mixed responses had lower overall survival and higher relapse rates compared to patients with a uniform response. This finding suggests that the presence of resistant tumors within the same patient may significantly impact disease progression.
However, the study also showed that patients who received additional local therapies, such as radiotherapy or surgery targeting resistant lesions, had improved clinical outcomes. Radiotherapy, in particular, has been shown to enhance immune response by inducing the release of tumor neoantigens and increasing T-cell infiltration into the tumor microenvironment.
These findings suggest that combining immunotherapy with local therapeutic strategies could be key to optimizing outcomes in patients with mixed responses, allowing for targeted treatment of non-responsive lesions.
Clinical Implications and the Future of Personalized Immunotherapy
The variability in immunotherapy response highlights the need for personalized strategies to maximize treatment efficacy. Tumor genetic characterization and tumor microenvironment analysis should be incorporated into clinical decision-making to predict which lesions are more likely to respond to immunotherapy and which may require complementary approaches.
The use of advanced technologies such as single-cell sequencing (scRNA-seq) and detailed tumor microenvironment (TME) analysis will enable more precise patient selection for immunotherapy. Additionally, combining systemic treatments with localized interventions, such as targeted radiotherapy, could optimize outcomes in patients with mixed responses.
In the future, developing new strategies to convert cold tumors into inflamed tumors could increase immunotherapy response rates. Modulating the tumor microenvironment through immune-modulating drugs, gene therapies, or therapeutic vaccines represents a promising avenue in overcoming immune resistance in cancer.
The study demonstrates that tumor heterogeneity and variability in immune infiltration are critical determinants of immunotherapy response. The coexistence of different tumor clones within the same patient can lead to mixed responses, posing a significant clinical challenge.
Combining immunotherapy with local treatments, such as radiotherapy or surgery, appears to be an effective strategy to improve survival in these patients. As we move forward in the era of precision oncology, integrating tumor genetics, immunology, and combination therapies will be essential for optimizing cancer treatments and improving patient outcomes.
Main Reference:
Morinaga, T., Inozume, T., Kawazu, M., Ueda, Y., Sax, N., Yamashita, K., Kawashima, S., Nagasaki, J., Ueno, T., Lin, J., Ohara, Y., Kuwata, T., Yukami, H., Kawazoe, A., Shitara, K., Honobe-Tabuchi, A., Ohnuma, T., Kawamura, T., Umeda, Y., Kawahara, Y., … Togashi, Y. (2022). Mixed Response to Cancer Immunotherapy is Driven by Intratumor Heterogeneity and Differential Interlesion Immune Infiltration. Cancer research communications, 2(7), 739–753. https://doi.org/10.1158/2767-9764.CRC-22-0050
Other References:
Schreiber, R. D., Old, L. J., & Smyth, M. J. (2011). Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science (New York, N.Y.), 331(6024), 1565–1570. https://doi.org/10.1126/science.1203486
Zou, W., Wolchok, J. D., & Chen, L. (2016). PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Science translational medicine, 8(328), 328rv4. https://doi.org/10.1126/scitranslmed.aad7118
Topalian, S. L., Drake, C. G., & Pardoll, D. M. (2015). Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer cell, 27(4), 450–461. https://doi.org/10.1016/j.ccell.2015.03.001


