For a long time, surgery has been the standard treatment for most solid tumors. Its primary goal is to remove as many malignant cells as possible, reducing the tumor burden and, in many cases, achieving disease remission. However, while surgery can be effective in removing the primary tumor, it does not always guarantee the complete eradication of cancer. One of the biggest challenges is that the surgical procedure, rather than being a definitive solution, can create conditions that favor metastatic dissemination and tumor recurrence, ultimately compromising long-term treatment success.
One of the most critical issues with surgery is postoperative immunosuppression. During and after the procedure, the body enters a physiological response state that includes the release of inflammatory mediators, increased oxidative stress, and alterations in immune activity. This phenomenon can reduce the immune system’s ability to detect and eliminate residual tumor cells, allowing some to escape and form metastases in other organs. This explains why many patients experience cancer relapse months or years after surgery, even when it appeared that the disease had been completely eliminated.
Surgery can also facilitate the dissemination of tumor cells into the bloodstream. During the procedure, some cancer cells may detach from the primary tumor and travel through the circulatory system, lodging in other organs and forming new metastatic foci. Additionally, the postoperative tissue healing process involves the release of growth factors such as VEGF (Vascular Endothelial Growth Factor), which, while necessary for tissue regeneration, can also promote the proliferation of residual tumor cells and the formation of new blood vessels within microtumors.
Given these challenges, intratumoral neoadjuvant immunotherapy has emerged as a promising strategy to improve surgical outcomes in oncology. Its goal is to prepare the immune system before the surgical procedure, allowing it to recognize and attack the tumor more effectively. A recent study published in Cancer Letters evaluated the efficacy of a combination immunotherapy called MBT(A) Therapy, administered directly into the tumor before surgery. The results were highly promising: the therapy not only reduced tumor progression but also prevented metastatic dissemination and generated long-lasting immune memory, significantly lowering the risk of recurrence.
This strategy is based on the combination of different immunostimulants designed to transform an immunosuppressive tumor microenvironment into an immunologically active one. The first component is Mannan-BAM (MB), an immune adjuvant that enhances antigen uptake and presentation by dendritic cells. These cells are essential for initiating and amplifying the adaptive immune response, enabling T cells to recognize and eliminate cancer more efficiently.
The second component consists of Toll-like receptor (TLR) ligands, which activate immune danger signals, induce the production of inflammatory cytokines, and promote immune cell infiltration into the tumor. This is crucial for overcoming the barriers that some solid tumors impose on immune system activity.
Finally, in the most advanced version of the therapy, anti-CD40 antibody (A) is included. This component targets the CD40 receptor, a key molecule in the activation of antigen-presenting cells and B lymphocytes. Its function is to enhance dendritic cell maturation, amplify T cell activation, and promote the formation of memory cells, ensuring that the immune system continues to recognize and attack any residual tumor cells after surgery.
The study evaluated this strategy in murine models of melanoma, pheochromocytoma, and metastatic breast carcinoma. The results demonstrated that neoadjuvant immunotherapy had a significant impact on preventing postoperative metastasis. In the pheochromocytoma model, MBTA completely prevented metastasis in 100% of treated subjects, while MBT (without anti-CD40) achieved an 86% reduction. In contrast, 60% of subjects in the control group developed metastases after surgery.
In breast carcinoma and melanoma models, the therapy significantly reduced the recurrence rate and improved survival. Additionally, immunological analyses revealed an increase in tumor-infiltrating cytotoxic T lymphocytes (CD8+ T cells), an upregulation of key cytokines such as IFN-γ and TNF-α, and a systemic activation of the immune response. One of the most important findings was that subjects treated with MBT(A) rejected tumor cell reintroduction without requiring further treatment, indicating that the therapy induced functional immune memory.
The impact of neoadjuvant immunotherapy is significant because it addresses a major issue in surgical oncology: the lack of an active immune response after tumor resection. Surgery is currently considered a curative tool, but in many cases, it is insufficient because it leaves open the possibility of recurrence. By stimulating a pre-surgical antitumor immune response, neoadjuvant immunotherapy allows the immune system to remain active and continue eliminating any residual cells that may have escaped during the operation.
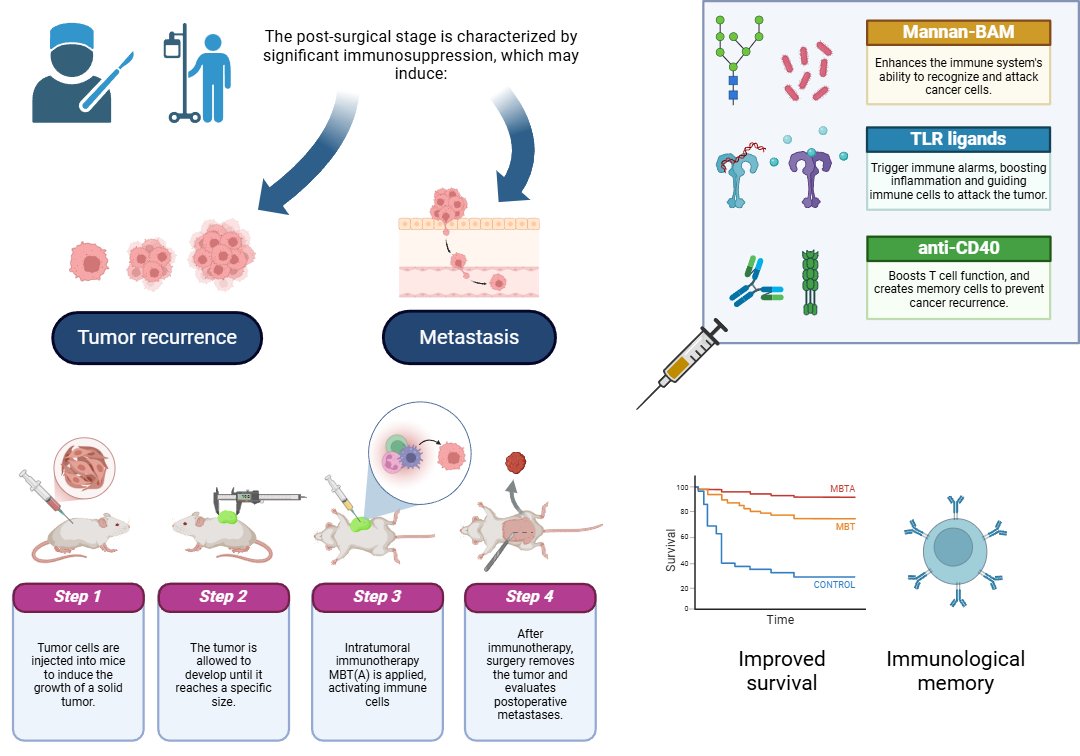
Figure 1. Intratumoral immunotherapy (MBT(A)) before surgery helps counteract post-surgical immunosuppression, reducing tumor recurrence and metastasis. By combining Mannan-BAM, TLR ligands, and anti-CD40, the treatment enhances immune activation, improving survival and immunological memory.
This approach could represent a paradigm shift in cancer treatment. Neoadjuvant immunotherapy not only reduces tumor burden before surgery but also protects the patient against relapse and metastatic dissemination. Additionally, by training the immune system, the therapy could enhance the effectiveness of subsequent treatments, such as systemic immunotherapy or chemotherapy, if needed.
Although this study was conducted in murine models, its results open the possibility of translating this strategy into clinical trials in humans. If validated in patients, this approach could be integrated into standard treatment protocols for tumors with a high risk of metastasis. The ability to reduce tumor burden, activate the immune response, and prevent cancer progression would make neoadjuvant immunotherapy a key component of the therapeutic arsenal against cancer.
Surgery will continue to be a fundamental pillar in cancer treatment, but it should not be seen as an isolated procedure. Combining surgery with pre-surgical immunological strategies could be the key to improving long-term survival and reducing tumor recurrence in cancer patients. Science continues to advance, and with it, the hope for more effective and personalized treatments that can offer better outcomes for those facing this disease.
Main Reference:
Uher, O., Hadrava Vanova, K., Labitt, R., Petrlakova, K., Ye, J., Wang, H., Masarik, M., Jakubek, M., Zenka, J., Zhuang, Z., & Pacak, K. (2025). Neoadjuvant intratumoral MBT(A) immunotherapy prevents distant metastases and recurrence in murine models. Cancer letters, 612, 217464. https://doi.org/10.1016/j.canlet.2025.217464
Other references:
Topalian, S. L., Forde, P. M., Emens, L. A., Yarchoan, M., Smith, K. N., & Pardoll, D. M. (2023). Neoadjuvant immune checkpoint blockade: A window of opportunity to advance cancer immunotherapy. Cancer Cell, 41(9), 1551–1566. https://doi.org/10.1016/j.ccell.2023.07.011
Caisová, V., Uher, O., Nedbalová, P., Jochmanová, I., Kvardová, K., Masáková, K., Krejčová, G., Paďouková, L., Chmelař, J., Kopecký, J., & Ženka, J. (2018). Effective cancer immunotherapy based on combination of TLR agonists with stimulation of phagocytosis. International Immunopharmacology, 59, 86–96. https://doi.org/10.1016/j.intimp.2018.03.038
Matsumoto, M., & Seya, T. (2008). TLR3: interferon induction by double-stranded RNA including poly(I:C). Advanced Drug Delivery Reviews, 60(7), 805–812. https://doi.org/10.1016/j.addr.2007.11.005


