How Beta-Blockers Enhance Oncolytic Virotherapy in Breast Cancer
When we think of cancer treatments, we often picture surgery, chemotherapy, radiotherapy, and immunotherapy. But what if our nervous system—specifically the sympathetic nervous system (SNS)—were silently influencing how tumors grow and resist treatment?
A recent study published in the Journal for ImmunoTherapy of Cancer sheds light on a surprising player in tumor resistance: stress-induced nerve signals. And even more surprising? A common medication, the beta-blocker, might hold the key to turning this disadvantage into an advantage during oncolytic virotherapy.
Oncolytic Viruses: Using Viruses to Destroy Tumors
Oncolytic herpes simplex virus (oHSV) therapy is a form of immunotherapy where a genetically modified virus is injected directly into tumors. The virus infects and destroys cancer cells while also alerting the immune system to mount a stronger response. While this approach has shown promise in hard-to-treat cancers like triple-negative breast cancer, its effects are often limited. Why?
The answer may lie in the nerves.
Stress Nerves Inside the Tumor?
The study found that injecting oHSV into tumors unexpectedly increased the growth of nerves—especially from the sympathetic nervous system, the same system involved in the “fight or flight” stress response. These nerves weren’t just present; they were releasing signals that promoted tumor growth, immune suppression, and treatment resistance.
This nerve growth also promoted the presence of perivascular macrophages, a type of immune cell that can block the activity of cytotoxic T cells. In short, the tumor was using the body’s own stress response to fight back.
What Happens When You Add a Beta-Blocker?
Researchers tested the combination of oHSV therapy with a beta-blocker—a medication commonly used to treat high blood pressure and heart conditions. The result? A significantly stronger antitumor response:
- More CD8+ T cells (the immune system’s cancer killers) infiltrated the tumor
- Nerve growth and the tumor’s immunosuppressive environment decreased
- Perivascular macrophages were reduced, and macrophages shifted toward a more inflammatory, tumor-fighting state
- T cell diversity increased, enabling a broader and more effective immune attack
Even more interesting, the combination therapy suppressed TGF-β, a molecule known to promote nerve growth and immune suppression within tumors.
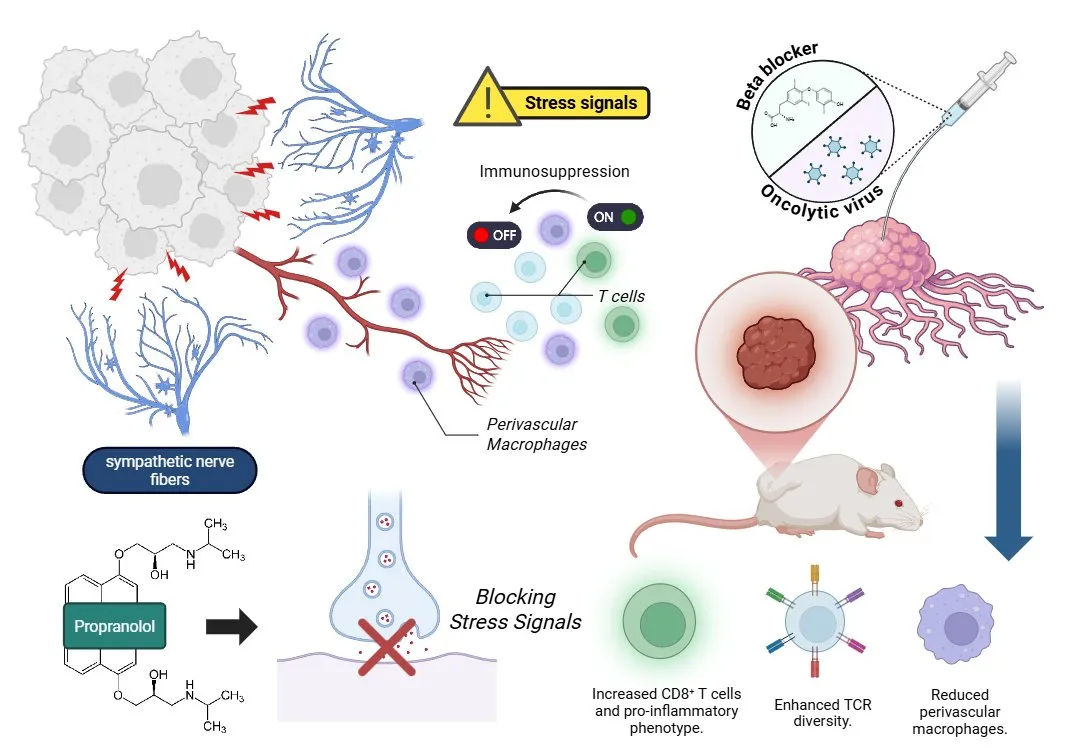
Figure 1. Sympathetic stress in the tumor microenvironment promotes immunosuppression through nerve-derived signals that activate perivascular macrophages and inhibit T cells. The combination of an oncolytic virus (oHSV) with the beta-blocker propranolol blocks these stress signals, enhancing CD8⁺ T cell infiltration, promoting a pro-inflammatory phenotype, increasing TCR diversity, and reducing perivascular macrophages.
Why This Matters
This research highlights a powerful concept: the nervous and immune systems are deeply connected in cancer biology. By understanding and manipulating this connection, we can make existing treatments—like oncolytic viruses—much more effective.
For patients with aggressive cancers such as triple-negative breast cancer, this could mean a new path to therapeutic success by simply adding a low-cost, already approved medication to the treatment plan.
Because sometimes, it’s not just about what we attack—but about how the body responds.
Main Reference
Kyritsi K, Pacholczyk R, Douglass E, et alβ-blocker suppresses both tumoral sympathetic neurons and perivascular macrophages during oncolytic herpes virotherapyJournal for ImmunoTherapy of Cancer 2025;13:e011322. doi: 10.1136/jitc-2024-011322. https://jitc.bmj.com/content/13/4/e011322
Other References
Pilipović, I., Vujnović, I., Stojić-Vukanić, Z., Petrović, R., Kosec, D., Nacka-Aleksić, M., … & Leposavić, G. (2019). Noradrenaline modulates CD4+ T cell priming in rat experimental autoimmune encephalomyelitis: a role for the α 1-adrenoceptor. Immunologic Research, 67, 223-240. https://link.springer.com/article/10.1007/s12026-019-09082-y
Globig, A. M., Zhao, S., Roginsky, J., Maltez, V. I., Guiza, J., Avina-Ochoa, N., … & Kaech, S. M. (2023). The β1-adrenergic receptor links sympathetic nerves to T cell exhaustion. Nature, 622(7982), 383-392. https://www.nature.com/articles/s41586-023-06568-6
Cosentino, M., Fietta, A. M., Ferrari, M., Rasini, E., Bombelli, R., Carcano, E., … & Lecchini, S. (2007). Human CD4+ CD25+ regulatory T cells selectively express tyrosine hydroxylase and contain endogenous catecholamines subserving an autocrine/paracrine inhibitory functional loop. Blood, 109(2), 632-642. https://ashpublications.org/blood/article/109/2/632/23562/Human-CD4-CD25-regulatory-T-cells-selectively


