For decades, surgery, chemotherapy, and radiation have been the cornerstone of cancer treatment. However, their side effects and impact on patients’ quality of life have driven the search for less invasive alternatives. Today, one of those alternatives is gaining momentum: immunotherapy. And a recent study has shown results so promising that they could completely change how we treat cancer.
In a groundbreaking clinical trial published in The New England Journal of Medicine and presented at the American Association for Cancer Research (AACR) conference, 12 patients with locally advanced rectal cancer and a specific genetic alteration called Mismatch Repair Deficiency (MMRd) were treated exclusively with an immunotherapy drug called dostarlimab.
For six months, the patients received intravenous infusions of the drug—no surgery, no chemotherapy, and no radiation. The results were astonishing: 100% of patients achieved complete clinical remission, meaning all detectable signs of the tumor disappeared. To date, none of them have relapsed, and all have avoided invasive procedures.
This finding not only generated scientific excitement but also sparked new hope for thousands of cancer patients seeking less aggressive and more effective options.
What Is MMRd and Why Does It Matter?
Mismatch Repair Deficiency (MMRd) refers to a malfunction in the cell’s system for fixing errors that occur during DNA replication. Under normal conditions, genes like MLH1, MSH2, MSH6, and PMS2 detect and correct these mistakes. When this system fails—whether due to inherited mutations or acquired damage—cells accumulate mutations more rapidly.
This increases the risk of cancer, but also creates a therapeutic advantage: tumors with MMRd produce so many mutations that they become more visible to the immune system, especially when aided by drugs like dostarlimab.
MMRd can occur in various cancers, either as part of a hereditary condition like Lynch syndrome, or spontaneously in patients with no family history. Today, molecular testing for MMRd or high microsatellite instability (MSI-H) is essential to identify which patients are most likely to benefit from immunotherapy.
Dostarlimab: Unleashing the Body’s Natural Defenses
Dostarlimab is a humanized monoclonal antibody that blocks the PD-1 protein, an immune checkpoint that cancer cells exploit to avoid being attacked. By inhibiting this pathway, the drug reactivates T cells, allowing them to recognize and destroy malignant cells.
It is administered through intravenous infusion, meaning the drug is delivered directly into a vein through an IV, ensuring rapid distribution throughout the body and interaction with the immune system.
Dostarlimab has shown particularly promising results in tumors with MMRd, and it has a favorable safety profile, with most side effects being mild and manageable compared to traditional chemotherapy.
Beyond Rectal Cancer: Other Cancers Where It Works
While the rectal cancer trial made headlines, dostarlimab is also being evaluated in other tumor types with encouraging results:
Endometrial cancer: In the GARNET trial, dostarlimab showed a 43.5% response rate in patients with advanced disease and MMRd.
Gastric and metastatic colorectal cancers: When combined with other therapies, it achieved disease control rates of up to 65% in early studies.
Prostate cancer with MMRd: Though rare, objective responses were seen in around 40% of patients in phase I/II trials.
Other solid tumors such as pancreatic, biliary, and lung cancers are being investigated in ongoing clinical trials, with early promising data in patients with high tumor mutational burden.
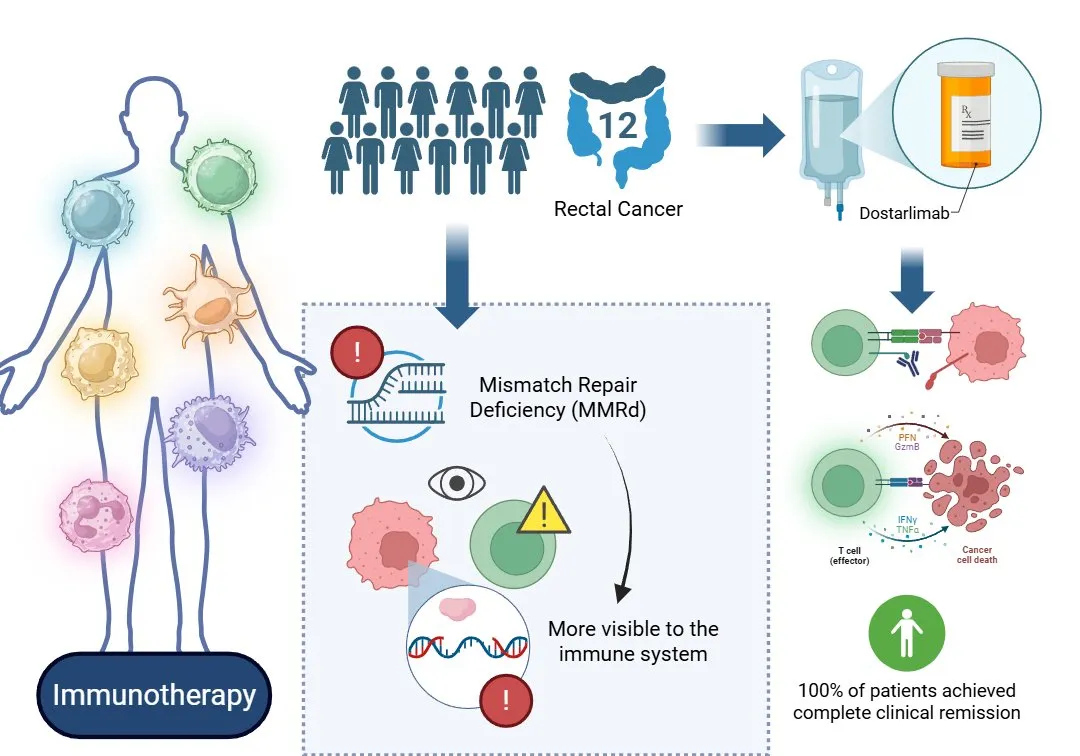
Figure 1. Dostarlimab helped the immune system target MMRd rectal tumors, leading to 100% remission without surgery, chemo, or radiation.
Regulatory Approvals and Clinical Use
The U.S. Food and Drug Administration (FDA) approved dostarlimab in 2021 for treating advanced endometrial cancer with MMRd in patients who no longer respond to chemotherapy. It also received “Breakthrough Therapy” designation, expediting its evaluation for broader indications.
In Europe, the European Medicines Agency (EMA) approved the drug for similar indications, and its use is expanding as more clinical data emerges.
A New Era in Oncology: Less Surgery, More Precision?
The story of dostarlimab represents what immunotherapy can achieve: treating cancer without the need for aggressive interventions. This approach not only promises better clinical outcomes, but also greatly improves quality of life for patients.
And most importantly, this is already happening. Immunotherapy is no longer experimental—it’s a real, accessible, and expanding tool in modern cancer treatment.
Immunotherapy Is No Longer the Future—It’s the Present
Dostarlimab is more than just another drug. It reflects a shift in mindset: training the immune system to defend itself, rather than attacking the body with broad, toxic treatments. This change represents a more strategic, personalized, and humane approach to medicine.
Immunotherapy is proving that it is indeed possible to fight cancer with less collateral damage, and its progress is unstoppable.
Main Reference:
Cercek, A., Lumish, M., Sinopoli, J., Weiss, J., Shia, J., Lamendola-Essel, M., El Dika, I. H., Segal, N., Shcherba, M., Sugarman, R., Stadler, Z., Yaeger, R., Smith, J. J., Rousseau, B., Argiles, G., Patel, M., Desai, A., Saltz, L. B., Widmar, M., Iyer, K., … Diaz, L. A., Jr (2022). PD-1 Blockade in Mismatch Repair-Deficient, Locally Advanced Rectal Cancer. The New England journal of medicine, 386(25), 2363–2376. https://doi.org/10.1056/NEJMoa2201445
Other References:
Emoghene, V. (2025, abril 29). Advance in immunotherapy show potential in cancer treatment. Rolling Out. https://rollingout.com/2025/04/29/advances-in-immunotherapy-cancer-surgery/
Cercek, A., Goodman, K. A., Hajj, C., Weisberger, E., Segal, N. H., Reidy-Lagunes, D. L., Stadler, Z. K., Wu, A. J., Weiser, M. R., Paty, P. B., Guillem, J. G., Nash, G. M., Temple, L. K., Garcia-Aguilar, J., & Saltz, L. B. (2014). Neoadjuvant chemotherapy first, followed by chemoradiation and then surgery, in the management of locally advanced rectal cancer. Journal of the National Comprehensive Cancer Network : JNCCN, 12(4), 513–519. https://doi.org/10.6004/jnccn.2014.0056
Chua, Y. J., Barbachano, Y., Cunningham, D., Oates, J. R., Brown, G., Wotherspoon, A., Tait, D., Massey, A., Tebbutt, N. C., & Chau, I. (2010). Neoadjuvant capecitabine and oxaliplatin before chemoradiotherapy and total mesorectal excision in MRI-defined poor-risk rectal cancer: a phase 2 trial. The Lancet. Oncology, 11(3), 241–248. https://doi.org/10.1016/S1470-2045(09)70381-X


