The human immune system acts like an intelligent army, constantly detecting and eliminating abnormal cells, including cancer cells. In recent years, immunotherapy has become one of the most exciting breakthroughs in medicine. However, not all patients respond to these therapies, and scientists have been trying to understand why.
A groundbreaking study published in Nature on February 6, 2025, led by Dr. Hideki Ikeda, Dr. Yosuke Togashi, and a large team of researchers from Japan, has uncovered a completely new strategy used by cancer cells: they transfer defective, mutated mitochondria into T cells, the very immune cells that are supposed to attack them. This mitochondrial sabotage weakens the immune response from the inside out.
Mitochondria are small structures within our cells responsible for producing energy. But their role goes beyond energy generation: they’re also involved in controlling inflammation, regulating cell death, and helping immune cells store memory of previous threats. Each mitochondrion contains its own DNA, called mitochondrial DNA or mtDNA, which is very prone to mutations. When a T cell receives damaged or defective mitochondria, it begins to malfunction. These dysfunctional mitochondria can also release harmful molecules known as reactive oxygen species (ROS), accelerating cellular aging and impairing immune function.
In this study, researchers examined tissue samples from patients with melanoma and non-small-cell lung cancer and made a surprising discovery: the tumor-infiltrating lymphocytes (TILs) had the exact same mitochondrial mutations as the cancer cells. This strongly suggested that the cancer cells were transferring their mitochondria to the immune cells. To confirm this, the scientists used advanced tracking techniques, including fluorescent proteins that light up mitochondria. These tools allowed them to observe, in real time, how mitochondria moved from cancer cells to T cells, both in laboratory cultures and in animal models.
There are two main ways this mitochondrial transfer happens. One is through tunneling nanotubes (TNTs), which are tiny, bridge-like structures that directly connect cells. The other involves extracellular vesicles (EVs), which are small, bubble-like particles that transport cellular components, including mitochondria, between cells. Normally, any foreign or damaged mitochondria entering a T cell should be destroyed by a process known as mitophagy (a kind of internal clean-up system for faulty mitochondria). However, the mitochondria that come from cancer cells are protected by a molecule called USP30. This molecule prevents their degradation, allowing them to remain inside the T cells and even replace the healthy mitochondria (a process known as homoplasmy), where all the mitochondria in a cell carry the same (mutated) genetic material.
The consequences for the T cells are significant. Their energy production drops dramatically, impairing key cellular functions. They become senescent, which means they age prematurely and lose their ability to divide. They begin to produce more ROS, further damaging their internal structures. Most importantly, they lose the ability to form immune memory, which makes it harder for them to respond to future threats. Even when they encounter cancer cells again, these T cells often fail to activate properly. In essence, cancer turns our own immune defenders into tired, unresponsive, and disoriented cells.
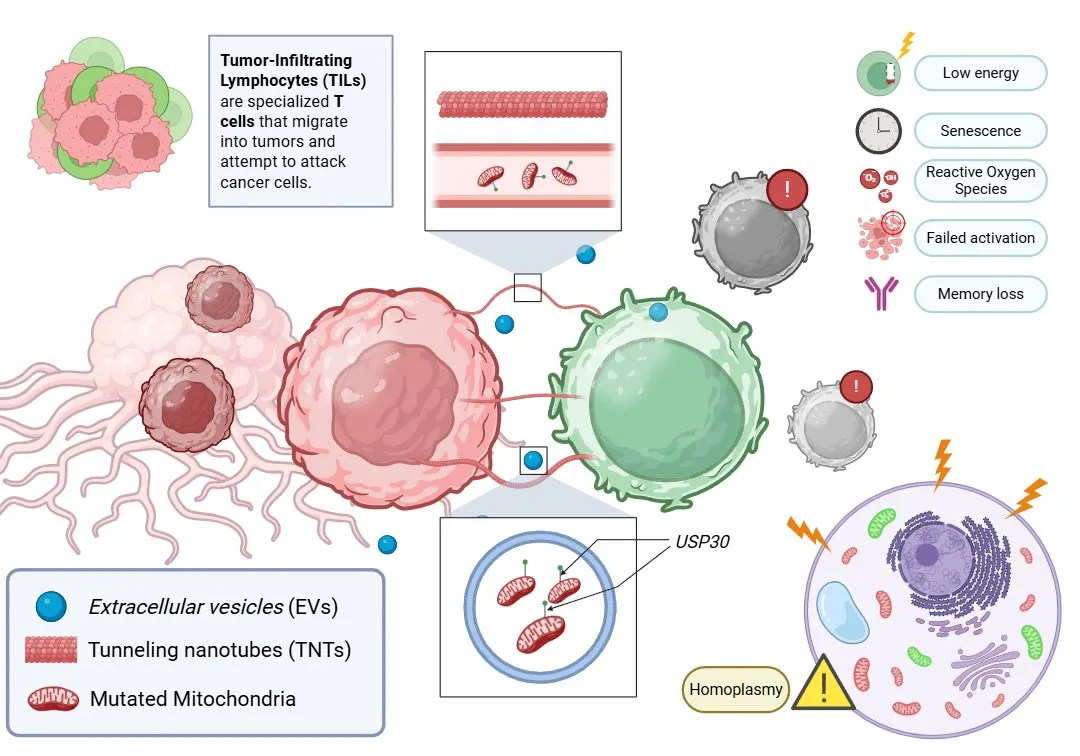
Figure 1: Mitochondrial transfer from tumor cells to TILs induces immune dysfunction.
Tumor cells transfer mutated mitochondria to tumor-infiltrating lymphocytes (TILs) via tunneling nanotubes (TNTs) and extracellular vesicles (EVs). These mitochondria evade degradation through USP30, leading to homoplasmy, impaired T cell activation, increased oxidative stress, senescence, and memory loss.
This discovery has major implications for cancer treatment. Modern immunotherapies (especially drugs that block immune checkpoints like PD-1) are designed to “wake up” exhausted T cells so they can resume their attack on the tumor. But this study shows that if the T cells have already been sabotaged by mitochondrial transfer from cancer cells, those therapies become much less effective. In animal experiments, tumors with mutated mitochondria continued to grow even when treated with PD-1 inhibitors. However, when mitochondrial transfer was blocked using a drug called GW4869, T cell function improved and the treatment response was better.
This study opens a new frontier in cancer research. It highlights the importance of looking beyond surface markers and immune signals—we must also consider what is happening inside the cell, especially within mitochondria. In the future, we may see new types of treatments that prevent the transfer of mutated mitochondria, block molecules like USP30 that protect those mitochondria from being broken down, reactivate mitophagy in affected T cells, and restore normal cellular metabolism. These strategies could make immunotherapy more effective, even for patients who currently don’t respond to it.
Main Reference:
Ikeda, H., Kawase, K., Nishi, T. et al. Immune evasion through mitochondrial transfer in the tumour microenvironment. Nature 638, 225–236 (2025). https://doi.org/10.1038/s41586-024-08439-0
Other References:
Schreiber, R. D., Old, L. J., & Smyth, M. J. (2011). Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science (New York, N.Y.), 331(6024), 1565–1570. https://doi.org/10.1126/science.1203486
DePeaux, K., & Delgoffe, G. M. (2021). Metabolic barriers to cancer immunotherapy. Nature reviews. Immunology, 21(12), 785–797. https://doi.org/10.1038/s41577-021-00541-y
Scharping, N. E., Menk, A. V., Moreci, R. S., Whetstone, R. D., Dadey, R. E., Watkins, S. C., Ferris, R. L., & Delgoffe, G. M. (2016). The Tumor Microenvironment Represses T Cell Mitochondrial Biogenesis to Drive Intratumoral T Cell Metabolic Insufficiency and Dysfunction. Immunity, 45(2), 374–388. https://doi.org/10.1016/j.immuni.2016.07.009


