One of the greatest challenges of immunotherapy is that not all patients respond equally. Many tumors, especially the so-called “cold” ones, are able to evade detection by the immune system and grow unchecked. This happens in part because the environment surrounding the tumor (known as the tumor microenvironment) is controlled by immune cells that—surprisingly—protect the cancer instead of fighting it. Among these, the most important are the regulatory T cells, or Tregs.
Under normal conditions, Tregs help maintain the balance of the immune system, preventing overreactions or autoimmunity (when the body attacks its own tissues). However, inside the tumor, these cells act as a shield for cancer, suppressing the inflammatory response that could eliminate it. This protective function depends on an intracellular signaling pathway known as the CBM complex, which includes three key proteins: CARMA1, BCL10, and MALT1.
This complex acts like a “master switch” that enables Tregs to exert their suppressive function. When the CBM is active, Tregs produce signals that inhibit inflammation and block the activation of other immune cells, such as cytotoxic T cells and macrophages, which are the main attackers of cancer cells.
In 2019, a group of researchers led by Mauro Di Pilato, Edward Y. Kim, and Thorsten Mempel from Massachusetts General Hospital and Harvard Medical School published an innovative study in the journal Nature. The aim of the study was to selectively block the CBM complex within the tumor, in order to deactivate the suppressive function of Tregs only in that environment, without affecting their role in the rest of the body. To achieve this, the researchers used two approaches:
– Genetic manipulation: They modified mice so that their Tregs could not activate CARMA1.
– Use of an inhibitory drug: They applied mepazine, a compound that inhibits the activity of MALT1, another key component of the CBM complex.
By doing this, the Tregs within the tumor lost their ability to suppress inflammation. But most notably, they not only stopped protecting the cancer—they completely changed their behavior.
When the CBM was blocked, the Tregs began to produce interferon gamma (IFNγ), a molecule they don’t normally secrete. IFNγ is a powerful pro-inflammatory cytokine that activates the rest of the immune system:
– It stimulates macrophages to become aggressive “tumor-eating” cells.
– It increases the expression of MHC molecules on tumor cells, making them more visible to T cells.
– It promotes the recruitment of cytotoxic T cells, the direct killers of cancer.
All of this transformed the tumor microenvironment—from a “silent,” tolerant space into an inflamed, hostile one full of alarm signals, which helped the immune system recognize and attack the tumor more effectively.
One of the most striking findings of the study was that, when CBM deactivation was combined with anti-PD-1 immunotherapy (a treatment that “releases the brakes” on the immune system), tumors that previously did not respond began to shrink or even disappear.
This is because many immunotherapies don’t work in tumors that have a suppressive environment, such as those with active Tregs. But if that environment is transformed from within—by making the Tregs switch roles and trigger inflammation—then the treatment has a much stronger foundation to work.
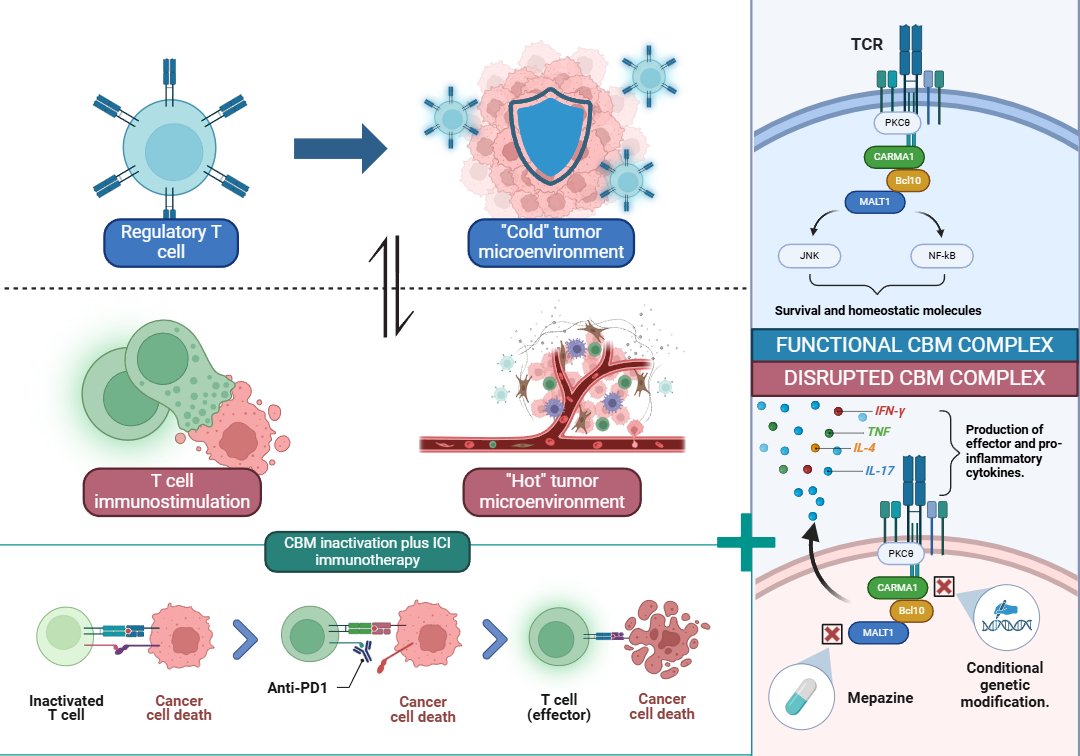
Figure 1. Inactivation of the CBM complex in Treg cells reprograms the tumor microenvironment from “cold” to “hot,” promoting the production of pro-inflammatory cytokines and enhancing anti-PD-1 immunotherapy. Disruption of the complex through mepazine or genetic modification enables effector T cell reactivation and tumor cell elimination.
The Role of Mepazine: An Old Drug, New Purpose
Mepazine is a medication that was once used as an antipsychotic, but in this study, it was repurposed for its ability to inhibit MALT1, one of the CBM complex proteins. When used, it achieved the same effect as genetic modification: destabilizing Tregs inside the tumor, prompting IFNγ production, activating local inflammation, and improving the immune response to therapy.
This finding is especially valuable because it shows that new drugs aren’t always necessary—sometimes, repurposing known and safe medications can be a fast and effective path toward new cancer treatments.
This study shows that cancer can be fought not only with drugs that attack the tumor directly, but also by reprogramming the environment that protects it. Changing the behavior of regulatory T cells (Tregs) from within the tumor allows the immune system to wake up and act, something that is crucial for patients who currently do not respond to immunotherapy.
Moreover, this strategy has an important added benefit: it’s safe. By acting specifically in the tumor, it does not disrupt the immune balance elsewhere in the body, which reduces the risk of serious side effects such as autoimmunity.
However, it’s important to clarify that this study was conducted in mouse models, so clinical trials in humans and further research are still needed to confirm its efficacy and safety before it can be applied in clinical practice. It’s a promising step, but still at an experimental stage.
Main Reference:
Di Pilato, M., Kim, E.Y., Cadilha, B.L. et al. Targeting the CBM complex causes Treg cells to prime tumours for immune checkpoint therapy. Nature 570, 112–116 (2019). https://doi.org/10.1038/s41586-019-1215-2
Other References:
Savage, P. A., Leventhal, D. S., & Malchow, S. (2014). Shaping the repertoire of tumor‐infiltrating effector and regulatory T cells. Immunological reviews, 259(1), 245-258.
Mellman, I., Coukos, G., & Dranoff, G. (2011). Cancer immunotherapy comes of age. Nature, 480(7378), 480-489.
Spranger, S., Spaapen, R. M., Zha, Y., Williams, J., Meng, Y., Ha, T. T., & Gajewski, T. F. (2013). Up-regulation of PD-L1, IDO, and Tregs in the melanoma tumor microenvironment is driven by CD8+ T cells. Science translational medicine, 5(200), 200ra116-200ra116.


