In recent years, colorectal cancer has become a growing threat to young adults, affecting people under 40, many of whom show no evident risk factors such as obesity, family history, or unhealthy lifestyles. This trend has puzzled oncologists and epidemiologists alike. However, a new study published in Nature on April 23, 2025, offers a surprising perspective that could change the way we understand this disease: its origin might lie in the gut—and more specifically, in a toxin called colibactin, produced by common bacteria acquired during childhood.
Colibactin is a genotoxic toxin secreted by specific strains of Escherichia coli, which naturally inhabit the human gut. Not all E. coli are harmful; in fact, many are part of a healthy gut microbiota. However, some strains possess a particular set of genes (known as the clb gene or pks genomic island) that enable them to synthesize colibactin. This toxin has the ability to interact directly with the DNA of colon cells, creating crosslinks and double-strand breaks that disrupt normal DNA replication and repair processes. If this type of damage is not properly corrected, it can lead to permanent mutations that, over time, accumulate and give rise to cancer.
The study, led by Dr. Ludmil Alexandrov at the University of California, San Diego, examined 981 colorectal tumor samples from patients across 11 countries. Using next-generation sequencing, the researchers analyzed the mutational signatures in each tumor—specific patterns of genetic damage that reveal what kinds of aggressors (like radiation, tobacco, or in this case, colibactin) have affected the DNA of the cancer cells.
The results were striking: mutational signatures linked to colibactin were more than three times more frequent in patients diagnosed before age 40 compared to those over 70. This strongly suggests that cases of colorectal cancer in young adults may have an early-life origin, possibly even during childhood.
But how does this early exposure occur? The researchers explain that colibactin-producing E. coli strains can be acquired in childhood through various routes: by drinking contaminated water, touching unclean surfaces, or putting dirty hands or objects in the mouth—as children often do when exploring their environment. These bacteria can also be transmitted through close contact with infected family members or by eating undercooked or improperly handled food. In countries with limited access to clean water and sanitation, exposure is more common, but it can also occur in developed countries if hygiene during early childhood is insufficient.
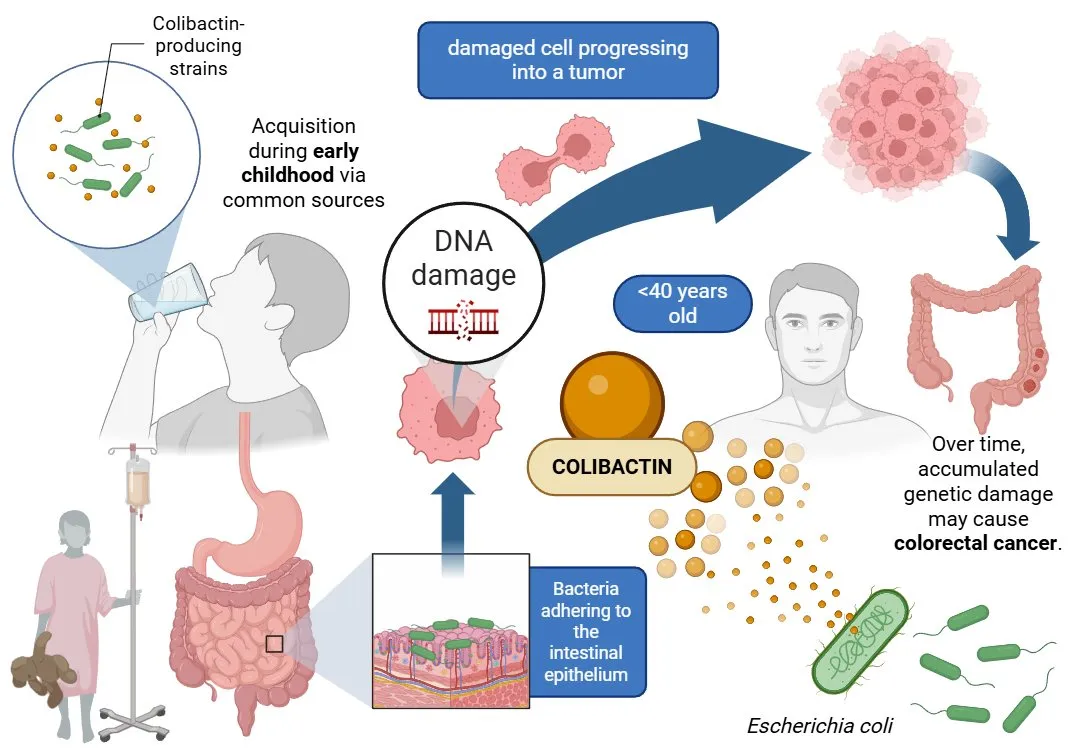
Figure 1. Early-life exposure to colibactin-producing E. coli can damage intestinal DNA, leading over time to colorectal cancer—especially in individuals under 40.
Once acquired, these strains can colonize the intestine and remain there persistently, becoming part of the microbiota without causing any visible symptoms. However, their chronic production of colibactin acts silently, causing occasional genetic lesions that the body may repair—until one goes uncorrected. Over the years, this accumulated damage may be the “trigger” that initiates tumor development.
This study represents a significant advance in understanding early-onset colorectal cancer. By demonstrating a direct molecular link between a bacterial toxin and the genetic profile of real tumors, scientists open new doors for prevention and early diagnosis. In the future, we may develop tests to detect these E. coli strains in the gut, especially in children or adolescents with a family history or digestive symptoms. Targeted therapeutic strategies could also be designed—such as probiotics that displace harmful strains, or even microbial vaccines.
Ultimately, this study highlights an uncomfortable truth: cancer doesn’t always arise from visible factors or adult lifestyle choices. Sometimes, it begins with invisible interactions between our cells and the microbes that have lived within us since childhood. Understanding and controlling those interactions could be key to stopping the silent epidemic of colorectal cancer in young people.
Main Reference:
Díaz-Gay, M., Dos Santos, W., Moody, S., Kazachkova, M., Abbasi, A., Steele, C. D., Vangara, R., Senkin, S., Wang, J., Fitzgerald, S., Bergstrom, E. N., Khandekar, A., Otlu, B., Abedi-Ardekani, B., de Carvalho, A. C., Cattiaux, T., Penha, R. C. C., Gaborieau, V., Chopard, P., Carreira, C., … Alexandrov, L. B. (2025). Geographic and age variations in mutational processes in colorectal cancer. Nature, 10.1038/s41586-025-09025-8. Advance online publication. https://doi.org/10.1038/s41586-025-09025-8
Other References:
Bray, F., Laversanne, M., Sung, H., Ferlay, J., Siegel, R. L., Soerjomataram, I., & Jemal, A. (2024). Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians, 74(3), 229–263. https://doi.org/10.3322/caac.21834
Brennan, P., & Davey-Smith, G. (2022). Identifying Novel Causes of Cancers to Enhance Cancer Prevention: New Strategies Are Needed. Journal of the National Cancer Institute, 114(3), 353–360. https://doi.org/10.1093/jnci/djab204
Kucab, J. E., Zou, X., Morganella, S., Joel, M., Nanda, A. S., Nagy, E., Gomez, C., Degasperi, A., Harris, R., Jackson, S. P., Arlt, V. M., Phillips, D. H., & Nik-Zainal, S. (2019). A Compendium of Mutational Signatures of Environmental Agents. Cell, 177(4), 821–836.e16. https://doi.org/10.1016/j.cell.2019.03.001


