The development of cancer vaccines has been a constant challenge in medicine, as the immune system often fails to effectively recognize tumor cells as threats. Unlike viruses and bacteria, which possess structures that are clearly foreign to the immune system, cancer cells originate from the host’s own body and can develop mechanisms to evade immune detection. Additionally, tumors can create an immunosuppressive microenvironment that inhibits the activity of key immune cells, such as dendritic cells and T lymphocytes. As a result, many cancer vaccines have struggled to induce immune responses that are strong and long-lasting enough to eliminate tumor cells.
To address this problem, scientists have been exploring innovative strategies, and one of the most promising approaches is the use of nanoparticles for in situ tumor vaccine delivery.
In a 2023 study conducted by Zhang and colleagues, an advanced strategy for cancer immunotherapy was proposed. The authors designed and developed the in situ vaccine presented in this work, representing a significant breakthrough in oncological research.
The In Situ Vaccination Strategy
The concept of an in situ vaccine involves converting the primary tumor into its own source of antigens, stimulating a specific immune response against cancer. In this study, researchers designed a system based on nanoparticles and a reactive oxygen species (ROS)-sensitive hydrogel to administer the vaccine directly into the tumor microenvironment. This strategy enables controlled drug release and efficient stimulation of dendritic cells, which are essential for immune activation.
Components of the In Situ Vaccine and Their Synergy
This innovative cancer vaccine comprises the following elements:
- CpG-P-ss-M Nanoparticles: Designed to adsorb tumor antigens and facilitate their transport to lymph nodes. These nanoparticles possess a special charge-reversal capability, optimizing dendritic cell activation. By shifting from a positive to a negative charge, they avoid rapid elimination from the body and enhance uptake by immune cells.
- ROS-Sensitive Hydrogel: Contains doxorubicin and CpG-P-ss-M nanoparticles. Its function is to release drugs in a controlled manner within the tumor microenvironment, promoting immunogenic cell death. The response to high ROS levels in tumors allows for sustained release, maximizing immune system stimulation.
- Whole-Cell Tumor Antigen: Generated in situ following doxorubicin-induced cell death, providing a reservoir of tumor-specific antigens. This ensures that the immune system learns to recognize multiple tumor markers, reducing the likelihood of immune escape.
- Anti-CTLA4 Antibody: Helps counteract tumor-induced immunosuppression, promoting a stronger immune response. It blocks immune checkpoint inhibitors that would otherwise limit T-cell activity.
- Losartan: Enhances immune cell infiltration into the tumor, allowing for a more effective immune response. Its action on the tumor microenvironment facilitates the arrival of activated T cells at the tumor site, increasing treatment efficacy.
The synergy among these components follows a well-coordinated sequence of events: doxorubicin induces immunogenic cell death and generates tumor antigens, which are captured by CpG-P-ss-M nanoparticles. These nanoparticles transport the antigens to lymph nodes, where they activate dendritic cells and, consequently, stimulate the proliferation of tumor-specific T cells. Simultaneously, the anti-CTLA4 antibody prevents excessive immune self-regulation, while losartan enhances immune cell infiltration into the tumor, maximizing the impact of the immune response.
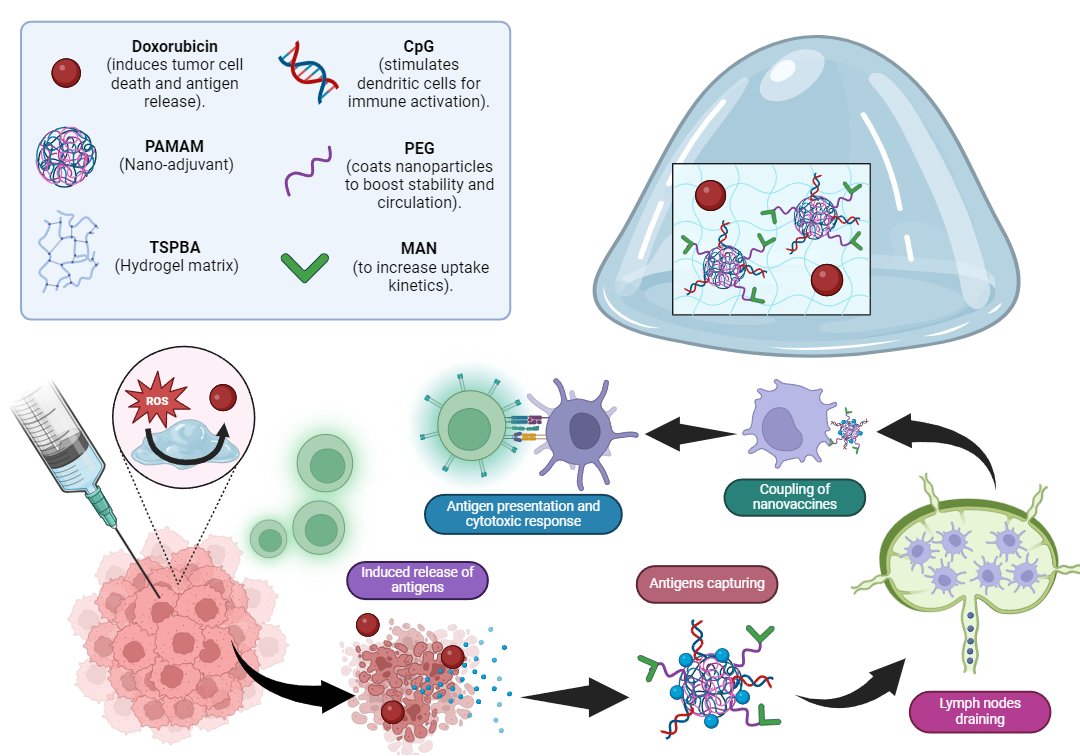
Figure 1. Schematic representation of the in situ tumor vaccine mechanism. The hydrogel matrix (TSPBA) releases its components in response to reactive oxygen species (ROS) in the tumor microenvironment. Doxorubicin induces tumor cell death and antigen release, while CpG stimulates dendritic cells for immune activation. PAMAM acts as a nano-adjuvant, and PEG enhances nanoparticle stability and circulation. MAN improves uptake kinetics. The released antigens are captured by nanovaccines, transported to lymph nodes, and presented to immune cells, triggering a cytotoxic immune response against the tumor.
Enhancing Immunotherapy with Checkpoint Inhibitors
One of the challenges in cancer immunotherapy is the presence of an immunosuppressive tumor microenvironment, which prevents the immune system from effectively attacking tumor cells. In this study, the in situ vaccine was combined with an anti-CTLA4 antibody and losartan, a drug that improves immune cell infiltration into tumors. This combination resulted in a 50% reduction in tumor growth, demonstrating the potential of this strategy to enhance cancer therapies.
Results and Future Prospects
The results obtained in murine breast cancer models show that this in situ vaccination strategy can generate a specific immune response against the tumor, activating cytotoxic T lymphocytes and promoting durable immunity. Additionally, a significant increase in immune cell infiltration into the tumor microenvironment was observed, suggesting that the vaccine not only induces a robust immune response but also modifies the tumor architecture to make it more susceptible to immune attack.
Studies revealed that mice treated with the combination of in situ vaccine, anti-CTLA4, and losartan exhibited improved survival rates compared to control groups. A reduction in metastasis formation was also observed, indicating that the immune response generated by the vaccine could have a prolonged effect in preventing tumor recurrence.
As research in nanotechnology and oncology advances, strategies like this could become key tools for personalized cancer treatment. The possibility of developing autologous vaccines from patients’ own tumors opens new opportunities for immunotherapy, reducing adverse effects and increasing treatment efficacy.
In conclusion, the use of nanoparticles in cancer vaccines represents a groundbreaking frontier in the fight against this disease. The combination of biomedical engineering, immunotherapy, and nanotechnology offers new hope for developing more effective and personalized treatments.
Main Reference:
Zhang, H., Zhang, Y., Hu, H., Yang, W., Xia, X., Lei, L., Lin, R., Li, J., Li, Y., & Gao, H. (2023). In Situ Tumor Vaccine for Lymph Nodes Delivery and Cancer Therapy Based on Small Size Nanoadjuvant. Small (Weinheim an der Bergstrasse, Germany), 19(33), e2301041. https://doi.org/10.1002/smll.202301041
Other References:
Guo, J., Liu, C., Qi, Z., Qiu, T., Zhang, J., & Yang, H. (2024). Engineering customized nanovaccines for enhanced cancer immunotherapy. Bioactive materials, 36, 330–357. https://doi.org/10.1016/j.bioactmat.2024.02.028
Li, Y., Tong, F., Wang, Y., Wang, J., Wu, M., Li, H., Guo, H., & Gao, H. (2024). In situ tumor vaccine with optimized nanoadjuvants and lymph node targeting capacity to treat ovarian cancer and metastases. Acta pharmaceutica Sinica. B, 14(9), 4102–4117. https://doi.org/10.1016/j.apsb.2024.06.003
Zha, Y., Fu, L., Liu, Z., Lin, J., & Huang, L. (2024). Construction of lymph nodes-targeting tumor vaccines by using the principle of DNA base complementary pairing to enhance anti-tumor cellular immune response. Journal of nanobiotechnology, 22(1), 230. https://doi.org/10.1186/s12951-024-02498-1


