Cancer remains one of the leading causes of mortality worldwide, with millions of new cases diagnosed each year. According to the World Health Organization (WHO), the number of cancer cases is expected to increase by 47% over the next two decades, posing a growing challenge for healthcare systems globally. Despite advances in treatments such as chemotherapy, radiotherapy, targeted therapies, and systemic immunotherapy, many tumors in advanced or metastatic stages remain difficult to treat due to resistance to conventional therapies and associated side effects.
Immunotherapy, which has revolutionized oncology in recent years, enhances the immune system’s ability to recognize and eliminate cancer cells. However, most current immunotherapies, such as immune checkpoint inhibitors (anti-PD-1, anti-CTLA-4) and adoptive cell therapy with CAR-T cells, are administered systemically, leading to severe side effects such as organ inflammation, hematological toxicity, and autoimmune reactions.
To overcome these challenges, a promising alternative has emerged: intratumoral immunotherapy, an approach aimed at increasing treatment efficacy while minimizing side effects.
What is Intratumoral Immunotherapy?
Unlike systemic immunotherapy, which is administered intravenously and circulates throughout the body, intratumoral immunotherapy directly injects immunotherapeutic agents into the tumor, which:
– Reduces systemic toxicity, as the drug remains concentrated at the tumor site without affecting healthy organs and tissues.
– Enhances local efficacy, ensuring higher bioavailability of therapeutic agents within the tumor microenvironment.
– Induces a systemic immune response, activating immune cells that can attack distant metastases—a phenomenon known as the abscopal effect.
This approach transforms the tumor into an “in situ vaccine”, meaning that the injection of immunotherapeutics stimulates the immune system to recognize and attack cancer cells throughout the body, including those located away from the primary tumor.
Advantages of Intratumoral Immunotherapy
Intratumoral immunotherapy offers multiple benefits compared to traditional approaches:
- Fewer systemic side effects: By concentrating on the tumor, toxicity to healthy organs is minimized, reducing the adverse effects commonly observed with systemic therapies, such as tissue inflammation, severe fatigue, and immune dysfunction.
- Greater local efficacy: Direct administration into the tumor allows for higher drug concentrations, improving effectiveness and reducing treatment resistance.
- Immune system stimulation: It can induce an adaptive immune response, allowing the immune system to continue targeting cancer cells even after treatment has ended.
- Lower costs: Smaller doses are required compared to systemic immunotherapy, potentially making treatment more accessible to a broader range of patients.
New Strategies in Intratumoral Immunotherapy
Intratumoral immunotherapy is not a single strategy but rather a collection of therapeutic approaches designed to optimize immune activation within the tumor. Research in this field has led to the development of several innovative methods that enhance the immune system’s ability to recognize and eliminate cancer cells while reducing systemic toxicity. Below are some of the most promising strategies currently being explored.
1.1. Oncolytic Viruses and Oncolytic Vaccines
Oncolytic viruses are genetically engineered viruses that selectively infect and destroy cancer cells while simultaneously stimulating the immune system to recognize tumor antigens. Unlike conventional treatments, these viruses replicate preferentially within tumor cells, causing their destruction and releasing tumor antigens that activate an anti-cancer immune response. A well-known example is Talimogene laherparepvec (T-VEC), a modified herpes simplex virus that has been approved for the treatment of advanced melanoma. T-VEC infects and lyses tumor cells, promoting the release of tumor-specific antigens and stimulating local and systemic immune responses. Other oncolytic viruses under investigation include adenovirus (VCN-01), reovirus, and measles virus, all of which have demonstrated promising results, particularly when combined with immune checkpoint inhibitors. These viruses serve as both cytotoxic agents and immune stimulators, enhancing the body’s ability to recognize and eliminate cancerous cells.
1.2. Peptide/Protein-Based Vaccines and Dendritic Cell Vaccines
Cancer vaccines aim to stimulate an immune response by introducing tumor-associated antigens that train immune cells to recognize and attack cancer cells. Peptide-based and protein-based vaccines contain small fragments of tumor antigens, such as neoantigens—proteins that arise from tumor-specific mutations—or recombinant proteins that mimic tumor markers. These vaccines induce an adaptive immune response by activating antigen-presenting cells, which, in turn, stimulate cytotoxic T cells to attack tumor cells displaying the same antigens.
Dendritic cell vaccines, on the other hand, take a more personalized approach by using the patient’s own immune cells to enhance antigen presentation. Dendritic cells, which are key players in immune activation, are extracted from the patient, loaded with tumor antigens in a laboratory, and then reinjected into the tumor. Once inside the tumor, these activated dendritic cells enhance T cell responses, leading to a more effective and targeted immune attack. These vaccines are being tested in various cancers and show great potential, particularly when used in combination with immune checkpoint inhibitors to further amplify the immune response.
- Intratumoral Immunomodulators
Intratumoral immunomodulators are compounds that enhance immune activation directly within the tumor microenvironment, counteracting the immunosuppressive conditions that often allow tumors to evade immune destruction. Some of the most studied immunomodulators include cytokines, such as IL-2 and IL-12, which promote T cell and natural killer (NK) cell activation. IL-2 enhances T cell proliferation and differentiation, while IL-12 facilitates the production of interferon-gamma (IFN-γ), a key cytokine involved in antitumor immunity.
Another emerging class of immunomodulators includes STING (Stimulator of Interferon Genes) agonists, which activate innate immune pathways to increase the production of pro-inflammatory cytokines and type I interferons. These agents create an inflammatory environment within the tumor, making it more visible to the immune system and improving T cell infiltration. Additionally, synthetic molecules designed to induce immunogenic cell death (ICD) are being investigated as a way to trigger a stronger and more durable anti-tumor immune response. By modifying the tumor microenvironment, these immunomodulators make the tumor more susceptible to immune-mediated destruction.
- Adoptive Cell Therapies
Adoptive cell therapy (ACT) is a personalized immunotherapy strategy that involves the modification and expansion of immune cells outside the body before reinfusing them into the patient. In an intratumoral context, ACT is being explored as a way to directly deliver potent immune cells into the tumor, bypassing the barriers that limit systemic immune cell infiltration.
One of the most promising ACT approaches is chimeric antigen receptor (CAR)-T cell therapy, where a patient’s T cells are genetically engineered to express receptors that specifically recognize tumor-associated antigens. Although CAR-T therapy has demonstrated great success in hematological cancers, its effectiveness in solid tumors has been limited due to the hostile tumor microenvironment. Intratumoral administration of CAR-T cells is being studied as a way to improve tumor infiltration, prolong cell persistence, and reduce off-target effects.
In addition to CAR-T cells, natural killer (NK) cells are being explored as another form of adoptive cell therapy. NK cells have the ability to recognize and eliminate tumor cells without prior antigen exposure, making them an attractive option for intratumoral administration. Moreover, dendritic cell-based therapies, which involve injecting antigen-loaded dendritic cells into the tumor, can significantly enhance T cell activation and lead to a robust and sustained anti-tumor immune response.
- Combined Therapies with Immune Checkpoint Inhibitors
Immune checkpoint inhibitors, such as anti-PD-1 and anti-CTLA-4, have transformed cancer treatment by reactivating exhausted T cells that would otherwise fail to attack tumors. However, some tumors develop resistance to these inhibitors due to their highly immunosuppressive microenvironment. Combining checkpoint inhibitors with intratumoral immunotherapy may enhance their effectiveness by increasing local immune activation. For example, intratumoral injections of checkpoint inhibitors ensure that the therapy remains concentrated at the tumor site, reducing systemic toxicity while amplifying the immune response. Additionally, these inhibitors work synergistically with other intratumoral treatments, such as oncolytic viruses and immunomodulators, to create a highly inflammatory tumor microenvironment that boosts T cell infiltration and persistence. These combination approaches are showing promising results in clinical trials and could significantly improve the outcomes of patients who do not respond to systemic checkpoint inhibition alone.
Controlled Drug Delivery Systems and Nanotechnology
One of the challenges in intratumoral immunotherapy is ensuring that therapeutic agents remain within the tumor for a sufficient duration to exert their full effect. Advances in nanotechnology, hydrogels, and injectable biomaterials are addressing this issue by improving drug stability, retention, and controlled release.
Nanoparticles are being developed to encapsulate immunotherapeutic agents, protecting them from degradation and allowing for more targeted delivery to tumors. These nanoparticles can be engineered to release their payload in response to specific tumor microenvironment signals, ensuring that the drugs are activated only at the tumor site. Hydrogels, which are gel-like materials capable of holding large amounts of water and therapeutic agents, are being designed for slow, sustained release of immunomodulators within tumors, prolonging their efficacy while reducing dosing frequency.
Injectable biomaterials, such as microspheres and polymer-based scaffolds, are also being investigated as a way to retain immunotherapeutic compounds within the tumor for extended periods, preventing premature clearance and enhancing local immune stimulation. These delivery systems have the potential to revolutionize intratumoral immunotherapy by improving the precision, duration, and safety of treatment.
Therapeutic Combinations to Enhance Efficacy
One of the most promising approaches in intratumoral immunotherapy is its combination with other therapeutic strategies:
Radiotherapy + Intratumoral Immunotherapy: Radiation can increase the presentation of tumor antigens, enhancing the immune response.
Chemotherapy + Intratumoral Immunotherapy: Induces immunogenic cell death, facilitating immune system activation.
Gene Therapies with CRISPR-Cas9: Being explored to improve the activity of immune cells injected into tumors.
Challenges and Future Opportunities
Despite its potential, intratumoral immunotherapy still faces several challenges:
- Tumor accessibility: Not all tumors are easily injectable, requiring advanced imaging techniques for precise administration.
- Tumor heterogeneity: Differences between tumors can affect treatment responses, necessitating personalized approaches.
- Durability of response: While intratumoral immunotherapy can induce strong responses, combining it with other strategies may be necessary to maintain long-term efficacy.
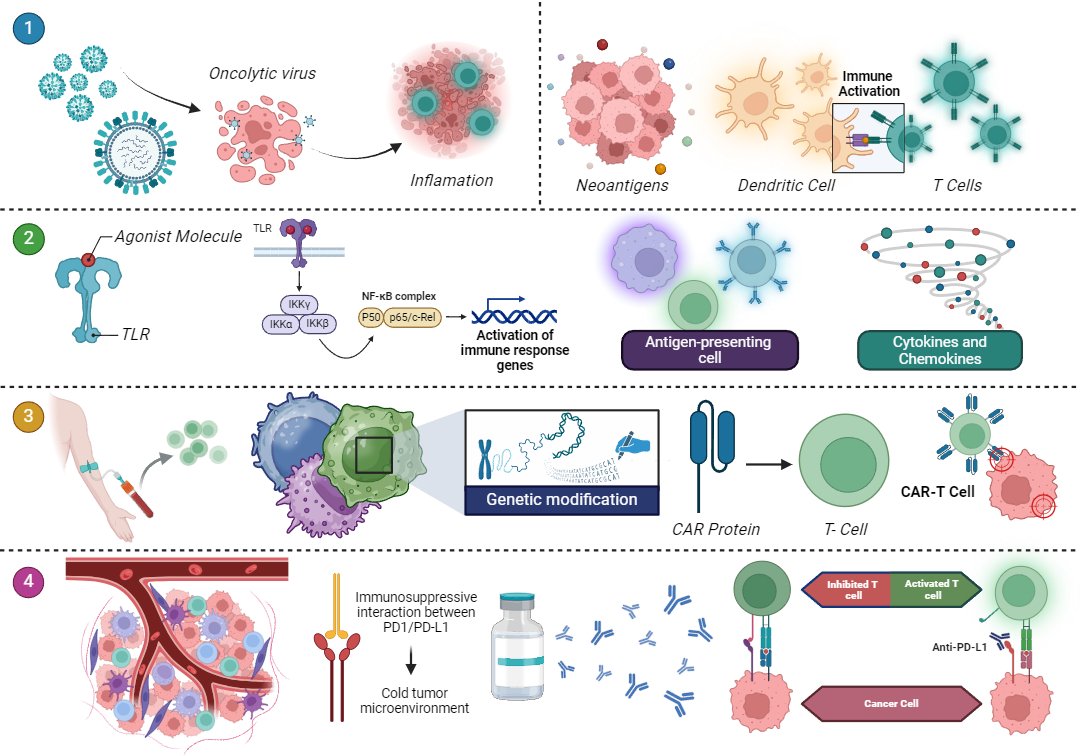
Figure 1. The figure illustrates different intratumoral immunotherapy strategies aimed at enhancing the immune response against cancer. Intratumoral cancer vaccines include oncolytic viruses, which destroy tumor cells and release neoantigens, promoting the activation of dendritic cells and T lymphocytes. Intratumoral immunomodulators, such as TLR agonists, activate the NF-κB pathway, inducing the production of cytokines and chemokines that amplify the immune response. Intratumoral adoptive cell therapies include CAR-T cell therapy, where T lymphocytes are genetically modified to recognize tumor antigens and attack cancer cells more effectively. Finally, intratumoral immune checkpoint inhibitors and monoclonal antibodies, such as anti-PD-L1 antibodies, block the PD-1/PD-L1 interaction, allowing T lymphocytes to reactivate and reducing tumor-induced immunosuppression. These strategies aim to transform the tumor microenvironment and improve the effectiveness of the immune response against cancer.
Conclusion
Intratumoral immunotherapy represents an innovative strategy with the potential to turn tumors into centers of immune activation. With advancements in adoptive cell therapies, oncolytic viruses, peptide/protein-based vaccines, and nanotechnology, the future of oncology is shifting toward more localized, effective, and less toxic treatments.
As clinical trials continue, this approach could transform the treatment of advanced cancer, offering new hope to patients worldwide.
Main Reference:
Skalickova, M., Hadrava Vanova, K., Uher, O., Leischner Fialova, J., Petrlakova, K., Masarik, M., Kejík, Z., Martasek, P., Pacak, K., & Jakubek, M. (2025). Injecting hope: the potential of intratumoral immunotherapy for locally advanced and metastatic cancer. Frontiers in immunology, 15. https://doi.org/10.3389/fimmu.2024.1479483
Other References:
Bray F, Laversanne M, Weiderpass E, Soerjomataram I. The ever-increasing importance of cancer as a leading cause of premature death worldwide. Cancer. (2021) 127:3029–30. doi: 10.1002/cncr.33587
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. (2021) 71:209–49. doi: 10.3322/caac.21660
Fares J, Fares MY, Khachfe HH, Salhab HA, Fares Y. Molecular principles of metastasis: a hallmark of cancer revisited. Signal transduction targeted Ther. (2020) 5:28. doi: 10.1038/s41392-020-0134-x


