In the challenging landscape of cancer treatment, not all tumors respond the same. Some, known as “cold tumors,” are particularly resistant to current immunotherapies because they manage to evade immune detection. This escape mechanism represents one of the greatest obstacles for the most advanced treatments, such as immune checkpoint inhibitors. Seeking a solution, a team of researchers led by Chiara Pastori and Robert Neal III, along with colleagues from Galvanize Therapeutics and the University of Pennsylvania, explored an innovative strategy: combining chemo-immunotherapy with an emerging technology called pulsed electric field (PEF).
The study, published in PLOS ONE in 2024, focused on a murine model of triple-negative breast cancer (4T1), considered highly aggressive and immunologically “cold.” The researchers applied a combination of chemotherapy (cisplatin), immunotherapy (anti-PD-1), and PEF to boost immune activation. This pulsed electric field technology does not generate heat or destroy essential cellular proteins, but instead induces a form of immunogenic cell death—a type of tumor cell death that stimulates the immune system to recognize and attack cancer cells. PEF works by applying very short, high-intensity electrical pulses directly to the tumor. These pulses alter the permeability of the cell membrane, creating transient pores that lead to cell death, while sparing the surrounding healthy tissue. Unlike thermal methods like radiofrequency or laser ablation, PEF acts more selectively and without collateral damage, making it an ideal tool for triggering antitumor immunity.
The experimental design included two main scenarios. In one, tumors were left intact to evaluate how effective the therapy was at controlling both the primary tumor and potential metastasis. In the other, tumors were surgically removed after treatment, simulating a neoadjuvant approach to assess its impact on residual or systemic disease. Flow cytometry, histological analysis of the lungs, and serum cytokine profiling were used to evaluate treatment outcomes.
The results were striking. In mice treated with the combination of PEF, chemotherapy, and immunotherapy, 44% survived long-term, whereas none of the mice in the group without PEF survived. Many also showed complete clearance of lung metastases—a phenomenon the authors attributed to the abscopal effect, where local tumor destruction leads to a systemic immune response that targets distant metastases. Furthermore, when tumor cells were re-implanted into mice that had overcome the disease, only those treated with the triple combination showed durable immunity, preventing the formation of new tumors.
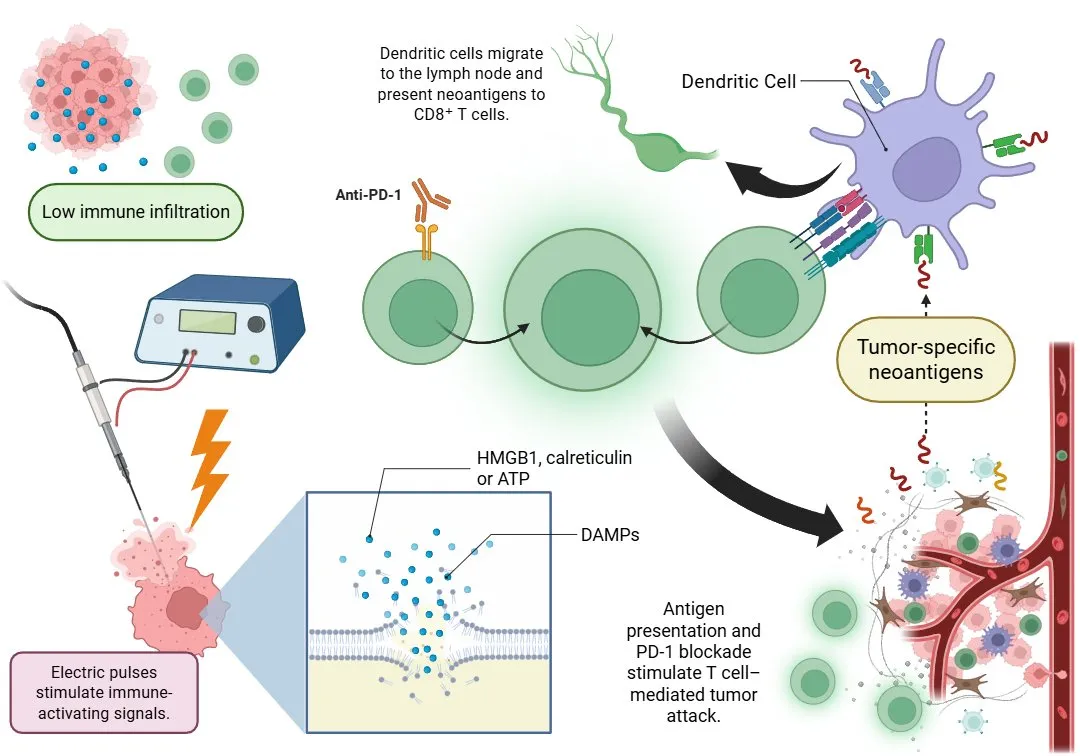
Figure 1. PEF technology enhances immune activation by turning cold tumors into targets for T cell–mediated attack, especially when combined with chemo-immunotherapy.
At the molecular level, the treatment significantly reduced the presence of cytokines associated with tumor progression and angiogenesis, such as IL-5, IL-13, VEGF, and CCL5. Meanwhile, the number of tumor-specific CD8+ T cells increased, reflecting effective activation of the immune system.
This study provides strong evidence that integrating pulsed electric field technology can significantly enhance the effectiveness of conventional immunotherapies. Not only does it improve local tumor control, but it also stimulates a systemic immune response capable of eliminating metastases and inducing long-term immune memory. Because it does not produce heat or damage adjacent tissues, PEF stands out as a safe, selective, and highly promising tool for combining with immunotherapy—especially in refractory or inoperable tumors.
This research suggests that a small electrical pulse can make a big difference in how we treat cancer. The integration of physical technologies with immunological therapies opens a new therapeutic frontier that could redefine cancer management in the coming years.
Main Reference:
Pastori, C., Nafie, E. H. O., Wagh, M. S., Hunt, S. J., & Neal, R. E., 2nd (2024). Neoadjuvant chemo-immunotherapy is improved with a novel pulsed electric field technology in an immune-cold murine model. PloS one, 19(3), e0299499. https://doi.org/10.1371/journal.pone.0299499
Other References:
Biemar, F., & Foti, M. (2013). Global progress against cancer-challenges and opportunities. Cancer biology & medicine, 10(4), 183–186. https://doi.org/10.7497/j.issn.2095-3941.2013.04.001
Esfahani, K., Roudaia, L., Buhlaiga, N., Del Rincon, S. V., Papneja, N., & Miller, W. H., Jr (2020). A review of cancer immunotherapy: from the past, to the present, to the future. Current oncology (Toronto, Ont.), 27(Suppl 2), S87–S97. https://doi.org/10.3747/co.27.5223
Robert, C., Karaszewska, B., Schachter, J., Rutkowski, P., Mackiewicz, A., Stroiakovski, D., Lichinitser, M., Dummer, R., Grange, F., Mortier, L., Chiarion-Sileni, V., Drucis, K., Krajsova, I., Hauschild, A., Lorigan, P., Wolter, P., Long, G. V., Flaherty, K., Nathan, P., Ribas, A., … Schadendorf, D. (2015). Improved overall survival in melanoma with combined dabrafenib and trametinib. The New England journal of medicine, 372(1), 30–39. https://doi.org/10.1056/NEJMoa1412690


