Glioblastoma (GBM) is the most aggressive brain tumor in adults, and despite advances in surgery, chemotherapy, and radiotherapy, it remains a disease with a discouraging prognosis. Glioblastoma stem cells (GSCs) pose a significant challenge as they have the ability to regenerate the tumor and resist conventional treatments. A new study published in Proceedings of the National Academy of Sciences (PNAS) 2025, led by Ling He and her team at the University of California, Los Angeles (UCLA), proposes an innovative strategy to tackle this disease: combining radiotherapy with forskolin to induce the differentiation of tumor cells into non-proliferative cell types.
The concept behind this study is to take advantage of a phenomenon that occurs after radiotherapy. It has been observed that radiation, in addition to destroying tumor cells, induces increased plasticity in surviving cells, leading them to a transient state of multipotency. This means that these cells may be more susceptible to reprogramming, which could be exploited to push them toward a non-tumorigenic cell fate. In this case, the researchers used forskolin, an adenylate cyclase activator that increases cyclic AMP (cAMP) levels in cells, promoting their differentiation into neurons and microglia.
To evaluate this strategy, the research team conducted experiments in cell cultures and animal models. First, they exposed glioblastoma cells derived from patients to radiation and then treated them with forskolin. Changes in the expression of neuronal and microglial markers were analyzed using single-cell RNA sequencing (scRNA-seq). In animal models, the effects of the treatment on tumor proliferation and mouse survival were tested.
The results were promising. The combination of radiotherapy and forskolin increased the expression of neuronal markers such as β3-tubulin and neurofilament light (NF-L), indicating that some tumor cells acquired neuronal characteristics. Additionally, an unexpected finding was that many cells adopted a phenotype similar to microglia, the immune cells of the brain. This change suggests that forskolin not only promotes neuronal differentiation but may also induce an immune response within the tumor.
Beyond cellular identity, the treatment had a significant impact on tumor cell proliferation. Cell cycle analysis showed an increase in cells arrested in the G1 phase, indicating a loss of cell division capacity. In animal models, the treatment reduced the number of glioblastoma stem cells and prolonged mouse survival. In patient-derived xenograft models, the median survival increased from 34 to 48 days, representing a significant improvement compared to conventional treatments.
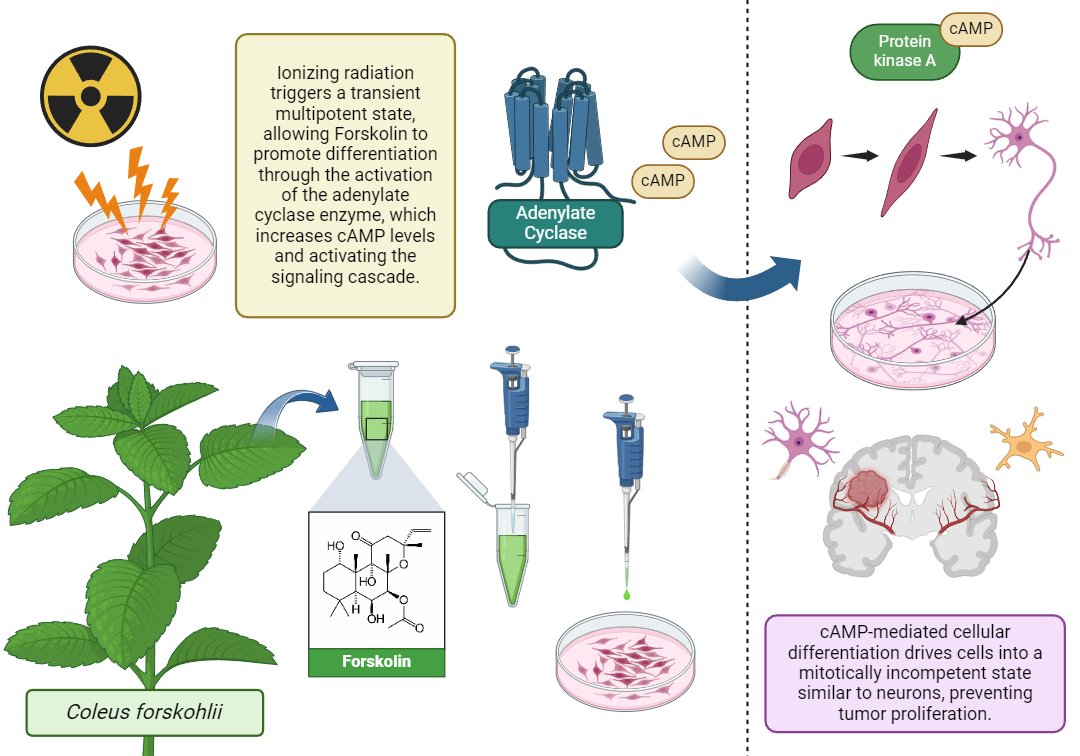
Figure 1. Mechanism of forskolin-induced cellular differentiation in glioblastoma: Ionizing radiation induces a transient multipotent state, allowing forskolin to activate adenylate cyclase and increase cAMP. This promotes cellular differentiation and blocks tumor proliferation.
Despite these encouraging results, the study’s authors acknowledge that challenges remain before this strategy can be tested in humans. One of the main questions is the durability of the observed effect. While the treatment delayed tumor progression, it did not completely eliminate it, suggesting that combinations with other differentiating agents or higher doses of forskolin may be necessary. Another challenge is the bioavailability of forskolin in the brain, as its ability to cross the blood-brain barrier still requires further study. Additionally, a deeper evaluation of long-term effects is needed since, although forskolin is used in Ayurvedic medicine, its impact on the brain in the context of glioblastoma is not yet fully understood.
This study provides evidence that the combination of radiotherapy and forskolin could open new possibilities in the fight against glioblastoma. The idea of inducing the differentiation of tumor cells into non-proliferative cell types represents an innovative approach that could complement existing therapies. Although further research is still needed to confirm the feasibility of this treatment in humans, this strategy could represent a significant breakthrough in the search for more effective therapies against this devastating cancer.
Main Reference:
L. He, D. Azizad, K. Bhat, A. Ioannidis, C.J. Hoffmann, E. Arambula, M. Eghbali, A. Bhaduri, H.I. Kornblum, & F. Pajonk, Radiation-induced cellular plasticity primes glioblastoma for forskolin-mediated differentiation (2025), Proc. Natl. Acad. Sci. U.S.A. 122 (9) e2415557122, https://doi.org/10.1073/pnas.2415557122
Other References:
Hemmati, H. D., Nakano, I., Lazareff, J. A., Masterman-Smith, M., Geschwind, D. H., Bronner-Fraser, M., & Kornblum, H. I. (2003). Cancerous stem cells can arise from pediatric brain tumors. Proceedings of the National Academy of Sciences of the United States of America, 100(25), 15178–15183. https://doi.org/10.1073/pnas.2036535100
Singh, S. K., Clarke, I. D., Terasaki, M., Bonn, V. E., Hawkins, C., Squire, J., & Dirks, P. B. (2003). Identification of a cancer stem cell in human brain tumors. Cancer research, 63(18), 5821–5828.
Bao, S., Wu, Q., McLendon, R. E., Hao, Y., Shi, Q., Hjelmeland, A. B., Dewhirst, M. W., Bigner, D. D., & Rich, J. N. (2006). Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature, 444(7120), 756–760. https://doi.org/10.1038/nature05236


