The gut microbiome, the community of bacteria and microorganisms living in our digestive system, is becoming a key player in cancer treatment. In recent years, research has shown that these bacteria are not only essential for overall health but also have a direct impact on how our body responds to advanced therapies such as immunotherapy.
Immunotherapy is a groundbreaking strategy that activates the immune system to fight cancer cells. However, not all patients respond to this approach in the same way. Recent studies have revealed that gut bacteria influence the effectiveness of these treatments. For example, certain types of bacteria, such as *Lactobacillus* and *Bifidobacterium*, seem to enhance the immune response, helping the body combat cancer more effectively. Conversely, a less diverse microbiome or one dominated by inflammatory bacteria could limit the benefits of immunotherapy or even exacerbate side effects.
This connection has led scientists to explore ways to modify the microbiome to improve outcomes in cancer patients. One of the most promising strategies is the use of probiotics, which are beneficial bacteria that can be consumed through supplements or fermented foods. Additionally, fecal microbiota transplants—a procedure in which gut bacteria from a healthy donor are transferred to a patient—have shown encouraging results in clinical trials. For instance, this technique has significantly improved responses to immune checkpoint inhibitors, a type of immunotherapy, in patients with advanced melanoma.
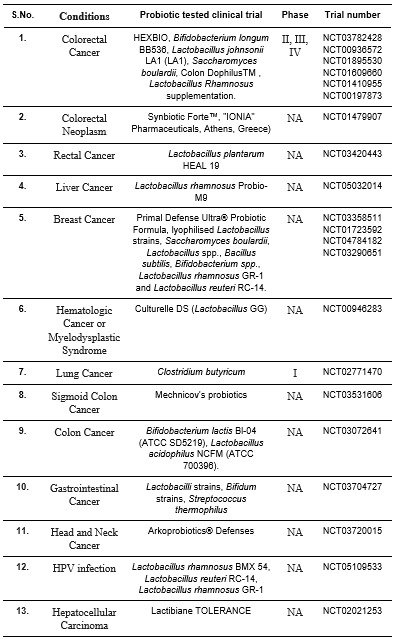
Table 1. The completed clinical trials testing the effect of probiotics on cancer immunotherapy (Modified from Singh et al. 2023).
Despite the progress, challenges remain. Not all bacteria have the same effect on all patients, and identifying the most effective combinations for specific types of cancer is crucial. Furthermore, more research is needed to understand how to balance the microbiome without causing unintended side effects.

Figure 1. The impact of the gut microbiome on antitumor immune responses, contrasting beneficial and detrimental microbial influences. It highlights how therapies like fecal microbiota transplantation (FMT), prebiotics, and postbiotics can modulate the microbiome to improve immune function and enhance cancer treatment outcomes.
The future of oncology may lie in personalizing the gut microbiome. Envisioning a treatment that combines immunotherapy with microbiome optimization is not just a promising idea—it’s a real possibility already being explored in laboratories and clinical trials. In the meantime, these discoveries offer new hope to those battling cancer, underscoring that we are not alone in this fight; even the smallest microorganisms can become powerful allies.
If you’re interested in learning more about how these innovations could apply to your treatment or that of a loved one, consult your doctor. Science is advancing rapidly, and with it, hope continues to grow.
Main Reference:
Singh, A., Alexander, S. G., & Martin, S. (2023). Gut microbiome homeostasis and the future of probiotics in cancer immunotherapy. Frontiers in immunology, 14, 1114499. https://doi.org/10.3389/fimmu.2023.1114499.
Other References:
Pitt, J. M., Vétizou, M., Waldschmitt, N., Kroemer, G., Chamaillard, M., Boneca, I. G., & Zitvogel, L. (2016). Fine-Tuning Cancer Immunotherapy: Optimizing the Gut Microbiome. Cancer research, 76(16), 4602–4607. https://doi.org/10.1158/0008-5472.CAN-16-0448
Derosa, L., Routy, B., Desilets, A., Daillère, R., Terrisse, S., Kroemer, G., & Zitvogel, L. (2021). Microbiota-Centered Interventions: The Next Breakthrough in Immuno-Oncology?. Cancer discovery, 11(10), 2396–2412. https://doi.org/10.1158/2159-8290.CD-21-0236
Suez, J., Zmora, N., Segal, E., & Elinav, E. (2019). The pros, cons, and many unknowns of probiotics. Nature medicine, 25(5), 716–729. https://doi.org/10.1038/s41591-019-0439-x


